Exam Prep
JEE Main, NEET 2022: How To Tackle Class 12 Inorganic Chemistry
Synopsis
Chemistry is actually one of the easiest sections to complete in the engineering and medical entrance exams, JEE Main and NEET. Inorganic Chemistry is usually a quick solve but it does require the candidate to be clear about concepts, know the periodic table of elements and have a strong grasp of everything in the Class 11 and Class 12 Chemistry textbooks. Questions in JEE Main and NEET, both conducted by the National Testing Agency (NTA), are seldom from outside the NCERT Chemistry Class 11 and Class 12 textbooks. Here are some quick tips on how to study Inorganic Chemistry for the exams.

Synopsis
Chemistry is actually one of the easiest sections to complete in the engineering and medical entrance exams, JEE Main and NEET. Inorganic Chemistry is usually a quick solve but it does require the candidate to be clear about concepts, know the periodic table of elements and have a strong grasp of everything in the Class 11 and Class 12 Chemistry textbooks. Questions in JEE Main and NEET, both conducted by the National Testing Agency (NTA), are seldom from outside the NCERT Chemistry Class 11 and Class 12 textbooks. Here are some quick tips on how to study Inorganic Chemistry for the exams.
When I ask my students what they think about Chemistry, they say they find it “difficult”, “confusing” and “full of exceptions”. Their response is understandable because in Chemistry, things don’t follow the usual trend or logic sometimes for one reason or the other. Physical chemistry seems difficult because of the myriad formulae and calculations involved. Organic Chemistry can be confusing if we try to memorise the reactions without understanding the reaction mechanisms and the various factors governing them. Studying Inorganic Chemistry is usually seen as demanding rote learning. However, an ideal way would be to understand the concepts thoroughly and practise hard so that one is at ease when it comes to exams.
The variations can seem overwhelming but Chemistry is actually one of the easiest subjects to crack in the Joint Entrance Examination (JEE Main and JEE Advanced) for Engineering admissions, and the National Eligibility cum Entrance Test (NEET UG) for admission to medicine. Almost everyone agrees that it is the least time-consuming part of the exam. Ideally one should be able to solve the Chemistry paper in under 45 minutes and devote the extra 15 minutes gained to the usually lengthy Mathematics paper and also for revisiting questions marked for review.
Even the most difficult questions of Inorganic Chemistry can be solved in under a minute while simpler ones take under 30 seconds. Then, if you don't know the answer to a particular problem, you can forget about it and move on to the next, unlike in Physics or Mathematics where you feel like you can solve it and end up wasting time on it. But with Inorganic, you know if you can solve a problem very quickly and don’t have to wonder. You quickly solve the problems you do know and leave the ones you don't. These two sections of Chemistry demand less time, compared to Physics or Mathematics but you must have a good grasp of Chemistry.
Also See,
- NEET, JEE Main: How To Tackle Class 12 Physical Chemistry
- This Is How You Tackle Organic Chemistry For JEE Main, NEET
- Career Options After 12th Science
Not all questions in these exams are difficult. The conducting authority, NTA for JEE Main and NEET, usually balances the difficulty level and you can often find very easy questions from the chapters which you find the most difficult. So, it is a good practice to not leave any chapter out of your preparation. Make sure you cover it at least to the level of the Class 12 Chemistry textbook of the National Council for Educational Research and Training (NCERT) which you will already require for your board exams.
Let us now look at how you can approach Inorganic Chemistry specifically. We will first classify the types of problems that are asked in the exams and then learn how to tackle them.
In Inorganic Chemistry, the types of problems asked can be broadly classified into the following types:
Structure-based and conceptual questions
Physical and chemical properties of elements in groups
Standard reaction-based problems
Chemistry: Structure-Based And Conceptual Problems
That Inorganic Chemistry is illogical and full of exceptions is a myth. Problems of the first type are logical and just require a deeper understanding of the bonding in different types of elements, and the different variables that can be predicted if we have a good understanding of the bonding. For example, questions related to bond parameters like bond angle, bond order, stability and hybridization. Must-do topics here are the structure of Oxoacids of Phosphorus, Sulphur and Chlorine, and the Oxides of Nitrogen. These topics are repeated cyclically in the examinations and just require an understanding of the bonding involved.
Coordination Compounds is another chapter from which similar conceptual questions are asked. Valence Bond Theory and Crystal Field Theory are the major topics we should focus on apart from Isomerism in Coordination Compounds from which the majority of the questions are framed.
Another important chapter in this subsection is Metallurgy which requires a deep understanding of various ores, their types and their extraction processes. The metallurgical extraction processes also require understanding of the thermodynamics involved in the reduction and the refining processes. Extraction of elements through leaching and Ellingham diagrams are some important topics in this section.
Jee Main, Neet Chemistry: Atomic , Physical Properties Of Elements
The next set of problems are on physical and chemical properties of elements and can be found in the following chapters:
Periodic classification of elements
Hydrogen
s block elements
p block elements
d and f block elements
The first step towards solving these problems is remembering the periodic table of elements and the location of each element in it. Many of these properties are periodic and vary in a similar fashion in the group or across a period. Tables are given in the NCERT textbooks related to the atomic and the physical properties of the elements. Focusing on these Tables will help you answer such problems.
We often ignore these tables or start to memorise the tables along with the data without attempting to understand. The trick lies in understanding that individual data need not be remembered and the overall data will fit a trend. So, just make a note of the elements which show an anomaly in the trend and revise them regularly.
The broader exceptions, can be attributed to a number of factors including shielding effect, inert pair effect and lanthanoid and actinoid contractions. Almost all of them are described in your NCERT textbook. A very important point to be noted is that the examiners very rarely venture into trends and exceptions beyond the scope of the NCERT textbook.
Another important class of questions is on the solubility of salts of different Oxoacids of the Alkali and the Alkaline Earth metals. The effect of heat on these compounds and their stability is also something which you should be paying attention to. These are in the NCERT Chemistry textbook as well and the examples given there are more than sufficient for your JEE Main or NEET preparation.
Class 12 Chemistry: Reaction-Based Problems
The third type of problems in Inorganic Chemistry are reaction-based in which we often need to predict the products obtained. These types of problems are the ones we usually don’t like as we tend to feel there are infinite reactions and most do not follow logic. The important thing to understand is that we are not supposed to remember each and every reaction occurring in the universe. The reactions which are asked in the exams are usually limited to a select few and are seldom from outside the NCERT textbook. We chase reference books for these reactions and end up creating a huge database and then struggle to keep track of them. You are only hurting yourself as most of these are not asked in the exams.
The questions asked in this section are usually related to standard reactions and are cyclically repeated as will be evident if you go through the previous years’ question papers. Some examples are preparation and properties of Potassium Permanganate, Potassium Dichromate, Hydrogen Peroxide, Nitric Acid and Sulphuric Acid, to name a few. Understanding these reactions, that is, whether these are redox reactions, acid base reactions or normal reactions like combination or displacement reactions, is important. We should focus on the behaviour of the reagent when they are subjected to certain reactions.
For example, let us consider the reaction of Copper metal with concentrated and dilute Nitric acid as shown below:
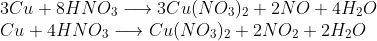
Copper reacts with both dilute as well as concentrated Nitric Acid and gets oxidised to the Cupric ions which are balanced by Nitrate ions to form Copper Nitrate. Nitric Acid when diluted, yields Nitric Oxide and when concentrated, yields Nitrogen Dioxide on reactions with different metals, typically, where they act as oxidising agents. Remembering this as a working principle is a lot easier than remembering the balanced chemical equation. Memorising balanced reactions is not advisable. Balancing these reactions will take about 60 seconds if you are thorough with the balancing of Redox reactions which is a part of your Physical Chemistry curriculum.
Reference Books For Inorganic Chemistry
We don’t need to chase “n” number of books and the NCERT is more than enough for you to prepare for the Inorganic Chemistry sections in JEE Main and NEET. Most reference books contain concepts and information that are more appropriate for undergraduate studies and the JEE and NEET seldom ask questions from them. More often than not, these books will confuse you and may steer you away from the subject.
Making your own notes for Inorganic Chemistry is very important as you will need to revise constantly. Make it a point to prepare complete notes from NCERT which is very well structured, with an easy-to-read flow of concepts and format. A good time to study Inorganic Chemistry is usually early mornings when our brains are fresh and our retention capacity at its peak.
Chemistry is much like life, where out of the number of factors involved, some dominate at some points and others at other times. Chasing the reasons behind these exceptions may not be good at this point, especially if your main goal is clearing the entrance exams for engineering and medicine. You will easily find lots of explanations on the internet. However, most of them are opinions and not facts, and in the process of applying them where they do not work, they will end up doing more harm than good.
Subscribe to Membership Plan
*Unlock all premium content and benefits:
Rate this Story
Latest Stories
MBBS In Russia? What You Must Know About Medical Colleges In Russia For Indian Students

Indian graduates who have completed MBBS from Russia cleared the FMGE exam and the success rate was 25%. Check here the Complete detail in this article.
By
7 min read ⋆ 16 Apr'24
MBBS In Russia: 25% Indian Graduates Cleared FMGE in 2022
7 min read ⋆ 16 Apr'24
Know the JEE Main 2023 cutoffs for NITs, IIITs and GFTIS
9 min read ⋆ 14 Apr'24
List of NITs Accepting Rank Below 2 Lakh in JEE Main
4 min read ⋆ 8 Apr'24
Careers360 helping shape your Career for a better tomorrow
250M+
Students
30,000+
Colleges
500+
Exams
1500+
E-Books
