- Chemistry
- Thermodynamics
- Graphical Comparison of Thermodynamic Processes
Graphical Comparison of Thermodynamic Processes
Thermodynamics is the area of physics dealing with heat and other types of energy. In particular, thermodynamics explains how thermal energy is transformed into and from other kinds of energy, and its interrelation with matter. Any thermodynamic process is a process of energy transfer within a system or between systems. The properties of a thermodynamic process are pressure, temperature, and volume. The state of the system at the present time is called the thermodynamic state of a system. An excellent example of a thermodynamic process is when food stays cool inside a refrigerator. A refrigerator removes the heat from the inside compartments and transfers it to the air on the outside.
This Story also Contains
- Constant Volume Process
- Work done during the process:
- Constant Pressure Process:
- Some Solved Examples
- Summary
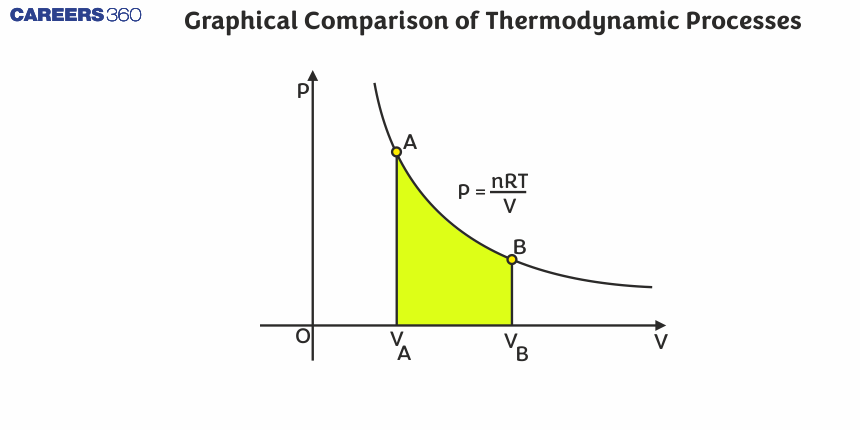
Constant Volume Process
Representation on P-V diagram:

This process is represented on the P-V diagram by a vertical straight line as shown in the figure, since V1=V2.
Work done during the process:
W=∫V1V2Pdv
But dv=0 for an isochoric process
∴W=0
Thus, work done during the constant volume process is zero which is also evident from the P-V diagram as no area is enclosed by the vertical line on the P-V diagram.
Constant Pressure Process:
Representation on P-V diagram:
During this process the pressure or the gas remains constant therefore it is represented by a horizontal line on the P-V diagram. See figure.

Work done during the process:
W=∫V1But P is constant.
∴ Work done =P∫V1V2dv=P(V2−V1)
A rectangle on P-V diagram represents the work done by the gas during the constant pressure process.
Recommended topic video on (Graphical Comparison of Thermodynamic Processes)
Recommended eBooks
Related eBooks and Sample Papers
Some Solved Examples
Example 1: One mole of an ideal gas expands from state X to Y by three paths 1, 2, and 3 as shown in the figure below. If W1, W2, and W3 are respective work done by ideal gas along the three paths then:

1)W1=W2=W3
2)W3>W1>W2
3) W3>W2>W1
4)W1>W2>W3
Solution
Work is a path function and not a state function. The area under P-V curve gives Work done. As far as the magnitude is concerned, work done by gas will be maximum in path 3 because the area under the curve is highest in the case of path 3. The second highest area is under path 2 and the least area is of curve 1.
Hence, Option number (3) is correct
Example 2: The magnitude of work done by a gas that undergoes a reversible expansion along the path ABC shown in the figure is

1) 489Correct)
2)85
3)58
4)54
Solution
W=(8−2)2+12((12−8)×(8−2))W=48
Example 3: What is the relation between the temperatures in the below graph which represents an isothermal expansion of gas at different temperatures?

1)T1>T2>T3
2)T2>T3>T1
3) T3>T2>T1
4)No relationship can be established
Solution
In the isothermal process, Temperature is constant.
Also, we know for an ideal gas, PV=nRT
As we can see T is constant, so we can say PV=k (here k is constant).
This equation of PV=k represents the equation of a hyperbola.
The higher the value of k the farther the curve is from the origin. So we can conclude that
T3>T2>T1
Hence, Option number (3) is correct
Example 4: Find out the magnitude of work (in kJ) done by one mole of an ideal gas for expansion.

1)-1
2)2
3) 6
4)60
Solution
Work is a path function and not a state function and the area under the P-V curve gives work.
So, work will be the total area of the Trapezium
∴∣ work |=12×( sum of parallel sides )×( distance between the parallel sides )∴∣ work |=12×6×20=60×10−5 bar − lit ∴∣ work ∣=60×105 bar − lit =60×105×10−3 bar −m3=6000 J
Thus, the magnitude of work done is 6 kJ
Example 5: One mole of an ideal monoatomic gas is subjected to changes as shown in the graph. The magnitude of the work done (by the system or on the system) is_________ J (nearest integer )

1) 6
2)7
3)4
4)6.5
Solution
I→II→ Isobaric II→III→ Isochoric III→I→ Isothermal WI−II=−1[40−20]=−20 Lit atm WII−III=0 WIV-I =2.303nRtlogV2 V1
=2.303PVlogV2 V1=2.303(1×20)log2=2.303×20×0.3010=13.818
W total =−20+13.818=(−6.182 lit atm )=6.182 lit atm
Summary
It's an interesting area of physics that deals mainly with the interaction of different kinds of energy, especially heat. How it describes the transformation of thermal energy into other forms of energy and how it influences the matter is quite interesting.
A thermodynamic process can be said to be a transfer of energy either within or between systems. The properties of the system in consideration are considered significant in these processes. The values of such properties at any given time describe the thermodynamic state of the system. An elementary example of a thermodynamic process could be the heating of water in a kettle. The heat from the surroundings—the stove—is transferred and absorbed by the kettle's system, which raises the temperature of the water.
25 Oct'25 - 15 Jun'26 (Online)
1 Nov'25 - 8 Jun'26 (Online)
15 Nov'25 - 4 Jun'26 (Online)
22 Dec'25 - 21 Jun'26 (Online)
29 Dec'25 - 6 Jun'26 (Online)

News and Notifications
Student Community: Where Questions Find Answers
