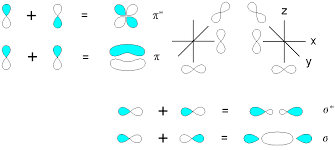
Sigma and Pi Bond - Difference, Definition, Structure, Properties, Uses, FAQs
Chemical bonds are very important in the field of chemistry. Chemical bonds are mainly classified into four categories they are; coordinate bonds, covalent bonds, ionic bonds, and hydrogen bonds. The bonds which are formed by sharing of electrons between two atoms are called covalent bonds and it is also known as a molecular bond. Sigma and Pi bonds are two different types of bonds that are formed in a covalent bond. The bond that is formed by the headways overlapping of atomic orbitals is called the sigma bond and is the strongest among the covalent bonds. Sigma bonds are denoted by the letter σ . And the bond which is formed by the lateral overlapping of atoms is called the pi bond is denoted by π.
This Story also Contains
- Write the difference between sigma bond and pi bond.
- Distinguishing features of sigma and pi bonds:
- Sigma Antibonding and pi Antibonding Orbitals
Also read -
Write the difference between sigma bond and pi bond.
Sigma bond and pi bond difference is shown in the below table.
Sigma bond | Pi bond |
Sigma bonds are formed by the headways overlapping of atomic orbitals. | Pi bonds are formed by the lateral overlapping of atomic orbitals. |
Sigma bonds are denoted by σ . | A pi bond is denoted by π. |
Sigma bond is the strongest bond. | The pi bonds are weaker. |
It can exist independently and free rotation is possible. | Pi bonds can be accessed only with the help of a sigma bond. |
It will determine the shape of the molecule. | Do not have a role in the shape of molecules. |
The overlapped orbitals formed by pure orbitals, hybrid orbitals and one hybrid orbital, and one pure orbital. | Overlapping orbitals are only pure orbitals. |
The sigma bond that can be formed between two atoms is one. | More than one pi bond can be formed between atoms. |
Distinguishing features of sigma and pi bonds:
The orbital overlapping of sigma and pi bonds is different, for a sigma bond formation headways overlapping between atomic orbitals will take place. The following images show the formation of a sigma bond between two atoms.
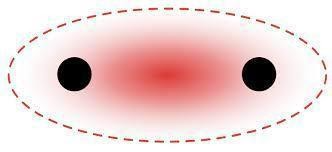
For a pi bond formation, the atomic orbitals are overlapped in a sideways manner. The following image shows the pi bond formation.
Sigma bond is usually defined for diatomic molecules and is formed by the headways overlapping between atomic orbitals. In the case of a Pi bond which is formed due to the lateral overlapping of orbitals and the bond is weaker than a sigma bond. A single bond always contains only one sigma bond and no pi bond is there eg. alkanes. In the case of multiple bonds like a triple and double bond, it contains pi bonds too. A double bond contains a one sigma bond and one Pi bond eg. alkenes. Similarly, in the case of a triple bond, there is one sigma bond and 2 Pi bonds are there eg. alkynes. The electron cloud in the case of the pi bond is unsymmetrical and in the case of the sigma bond is symmetrical.
Sigma Antibonding and pi Antibonding Orbitals
When an electron is present outside the region between two nuclei it is called an antibonding molecular orbital. The orbitals of two atoms overlap when they approach each other and thereby form a bond but in the case of antibonding a bond will not be formed. No overlapping of orbitals takes place; this is the only repulsion. Sigma antibonding is represented by a sigma star that is raised to an asterisk, σ*. Pi antibonding is represented by the pi star that is raised to an asterisk, π*.\
Also Read:
- NCERT solutions for Class 11 Chemistry Chapter 12 Organic chemistry- some basic principles and
techniques - NCERT Exemplar Class 11 Chemistry Solutions Chapter 12 Organic chemistry- some basic principles and
techniques - NCERT notes Class 11 Chemistry Chapter 12 Organic chemistry- some basic principles and techniques
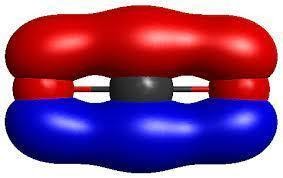
The Following Figure Shows the Formation of Antibonding Orbitals.
When there is a presence of extra electron like in the case of H-2 the extra electron will be present in the antibonding orbital. Thus thereby avoiding the extra electron interacting with other electrons. Antibonding orbitals have more energy compared to the bonding molecular orbitals. Only the bonding molecular orbitals participate in the bond formation. And the energy difference between two bonding molecular orbital and antibonding molecular orbital is high. The figure below shows the bonding and antibonding molecular orbital formation in the hydrogen molecule.
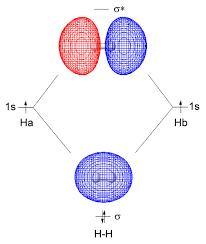
The bonding molecular orbital is formed by the combination of + and + part or – and – part of atomic orbitals and thereby forming a bond in between the two atoms. While the antibonding molecular orbital is formed by the combination of – and + part of atomic orbitals and thereby no bond formation is taking place. And for an antibonding molecular orbital the probability of finding an electron is low and correspondingly the electron density is low.
Also check-
- NCERT Exemplar Class 11th Chemistry Solutions
- NCERT Exemplar Class 12th Chemistry Solutions
- NCERT Exemplar Solutions for All Subjects
NCERT Chemistry Notes: