Surface Tension - Definition, Examples, Formula, Unit, Dimension, FAQs
Surface tension is an important concept in Class 11 Physics that explains why liquid surfaces behave like a stretched elastic membrane. Have you ever noticed water forming rounded droplets or insects walking on water? These phenomena occur due to surface tension, which is defined as the force acting per unit length on the surface of a liquid. This chapter, part of Properties of Solids and Liquids, is highly important for board exams and competitive exams like JEE Main and NEET. In this article, you will learn the definition of surface tension, formula (T = F/l), SI unit (N/m), dimensional formula, causes, temperature dependence, and solved numerical problems to strengthen your conceptual understanding and exam preparation.
This Story also Contains
- What is Surface Tension?
- What Causes Surface Tension?
- Examples of Surface Tension
- Solved Example Based On Surface Tension

What is Surface Tension?
Surface tension is the elastic tendency of a fluid surface which makes it acquire the least surface area.
If we draw an imaginary line on the free surface of the liquid as shown in the below figure.

Then Surface tension of a liquid is measured by the force acting per unit length on either side of an imaginary line.
So Surface tension of a liquid is given by
$T=\frac{F}{l}$
Where:
F→ force
l→ imaginary length
The direction of this force is perpendicular to the line and tangential to the free surface of the liquid.
It depends only on the nature of the liquid and is independent of the area of the surface or length of the imaginary line considered.
- It is a scalar quantity.
Unit of Surface Tension
- Force is measured in newtons ( N ).
- Length is measured in meters (m).
$T=\frac{\mathrm{N}}{\mathrm{~m}}=\mathrm{N} / \mathrm{m}$
SI Unit: $\mathrm{N} / \mathrm{m}$. - Dimensional Formula: $[T]=M T^{-2}$.
Example- Raindrops are spherical in shape because each drop tends to acquire minimum surface area due to surface tension, and for a given volume, the surface area of the sphere is minimum.
The surface tension of liquid decreases with the rise in temperature.
What Causes Surface Tension?
Surface tension is caused by intermolecular forces of attraction between the molecules of a liquid.
Inside a liquid, each molecule is surrounded by other molecules and is pulled equally in all directions. As a result, the net force on these molecules is zero.
However, molecules at the surface of the liquid do not have neighbouring molecules above them. Therefore, they experience a net inward force due to unbalanced cohesive forces. This inward pull makes the surface molecules contract and behave like a stretched elastic membrane.
Because of this effect, the liquid surface tends to minimize its surface area, forming spherical droplets and allowing small insects to walk on water.
Examples of Surface Tension
Surface tension can be observed in many everyday situations. Some common examples are:
Water droplets forming spheres
Rain drops and small water droplets form a spherical shape because surface tension minimizes the surface area.
Insects walking on water
Small insects like water striders can walk on water due to the thin “elastic-like” surface formed by surface tension.
Floating of a needle or razor blade
A small needle can float on water if placed carefully, even though it is denser than water.
Soap bubbles
Soap solutions form bubbles because surface tension pulls the liquid into a thin spherical film.
Capillary action
Water rises in thin tubes or plant stems due to surface tension along with adhesive forces.
Formation of drops from a tap
Water collects and forms drops before falling because surface tension holds the molecules together.
For More Information On Surface Tension, Watch The Below Video:
Solved Example Based On Surface Tension
Example 1: A drop of liquid of density ρ is floating half immersed in a liquid of density σ and surface tension 7.5×10−4Nem−1. The radius of drop-in will be :
(g=10 ms−2)
1) 15(2ρ−σ) 2) 15(ρ−σ) 3) 32(ρ−σ) 4) 320(2ρ−σ)
Solution:

B+T(2πR)=mgσ×2πR33 g+(2πR)=ϱ×2πR33 gσR2 g3+T=ϱR2 g37.5×10−2=R2 g3(ϱ−σ)R=32(ρ−σ)
Hence, the answer is option (3).
Example 2: A spherical soap bubble of radius 3 cm is formed inside another spherical soap bubble of radius 6 cm. If the internal pressure of the smaller bubble of radius 3 cm in the above system is equal to the internal pressure of another single soap bubble of radius r cm. The value of r is___________.
1) 2
2) 4
3) 3
4) 1
Solution:

Pi−P=4 Tr1P−Po=4 Tr2Pi−Po=4 Tr1+4 Tr2→(1)

Pi−Po=4 Tr→(2) From eq. (1) and (2) 4tr=4 Tr1+4 Tr2 (Given) 1r=1r1+1r2=13+16=918r=2 cm
Hence, the answer is option (1).
Example 3: A thin liquid film formed between a U- U-shaped wire and a light slider supports a weight of 1.5 $\times$10-2 N ( see figure) The length of the slider is 30 cm and its weight is negligible. The surface tension of the liquid film is:

1) 0.0125 Nm-1
2) 0.1 Nm-1
3) 0.05 Nm-1
4) 0.025 Nm-1
Solution:
The force due to the surface tension will balance the weight.

2TL=wT=w2L
Substituting the given values, we get
T=1.5×10−2 N2×30×10−2 m=0.025Nm−1
Hence, the answer is option (4).
Example 4: On heating water, bubbles being formed at the bottom of the vessel detach and rise. Take the bubbles to be spheres of radius R and making a circular contact of radius r with the bottom of the vessel. If r << R, and the surface tension of water is T, value of r just before bubbles detach is :(density of water is $\rho _w$ )

1) R22pwg3T
2) R2pwg6T
3) R2pwgT
4) R23pwgT
Solution:
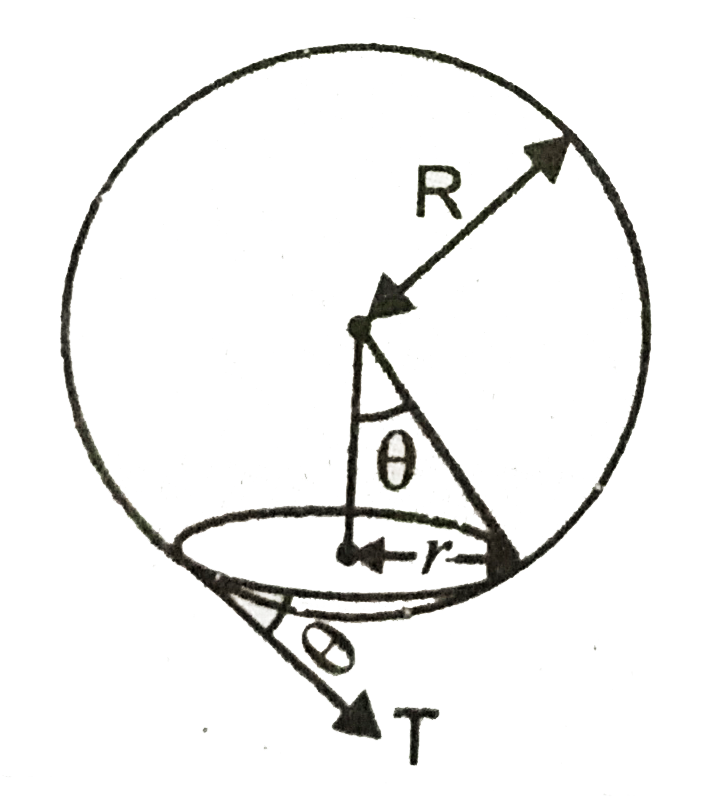
Here T is the surface tension and we know that surface tension,
T=Fl=F=Tl=T(2πr),
where F is the force due to surface tension and its vertical component will be T(2πr)Sinθ. .
Now The bubble will detach, if Buoyant force ≥ vertical component of surface tension force, so for the bubble to just detach:-
vertical component of surface tension force=buoyant force(or upthrust)
T(2πr)Sinθ=Vρwg⇒T(2πr)rR=43πR3ρwg⇒r2=2R4ρwg3T⇒r=R22ρwg3T
Hence, the answer is option (1).
Example 5: A soap bubble, blown by a mechanical pump at the mouth of a tube, increases in volume, with time, at a constant rate. The graph that correctly depicts the time dependence of pressure inside the bubble is given by :
1)
2)
3)
4) none of these
Solution:
Pressure Difference in Water Droplet -
$\Delta P=\frac{2 T}{R}$
wherein:
T- Surface tension
R- Radius
$V=c t^{4 / 3}=\frac{4}{3} \pi R^3$
$
R=K t^{1 / 3}
$
$
P=P_0+\frac{4 T}{K t^{1 / 3}}
$
$
P=P_0+C t^{-1 / 3}
$
Hence, the answer is option (4).
Frequently Asked Questions (FAQs)
Raindrops are spherical in shape due to the presence of cohesive forces between liquid molecules and the surface tension of the liquid.
Formula of surface tension is T = F/L
The unit of surface tension is Newton/meter(N/m).
The dimensions of surface tension are [MT^-2]
It is zero at boiling point.




