Swarts Reaction - Mechanism, Application, Examples, FAQs
Have you ever thought about how scientists replace one type of atom in a molecule with another to create new compounds? What happens when the halogen atom in an alkyl halide is exchanged for a fluorine atom? The answer to these questions is Swart's reaction. It is a process of converting alkyl chloride or alkyl bromide to form alkyl fluoride. It is an organic reaction. The reaction got its name after Frederic Jean Edmond Swarts, who reported and explained this reaction for the first time in 1892.
This Story also Contains
- Swart's Reaction
- Swarts Reaction Mechanism
- Applications of the Swarts Reaction
- Finkelstein Reaction
- Some Solved Examples
In this article, we cover the Swarts reaction class 12 chapter, haloalkanes and haloarenes. Which is very important for boards and JEE Mains Exam, NEET Exam, and many others.
Swart's Reaction
The best method for the preparation of alkyl fluorides is the Swarts reaction. This reaction is done by heating alkyl chloride or alkyl bromide in the presence of heavy metal fluorides. We can use mercurous fluoride or silver fluoride in the form of heavy metal fluorides. If we use light metal fluorides, then the reaction will continue in the forward direction, but the product formed will be much less. This is simply a fluorination reaction.
Example of Swart's reaction
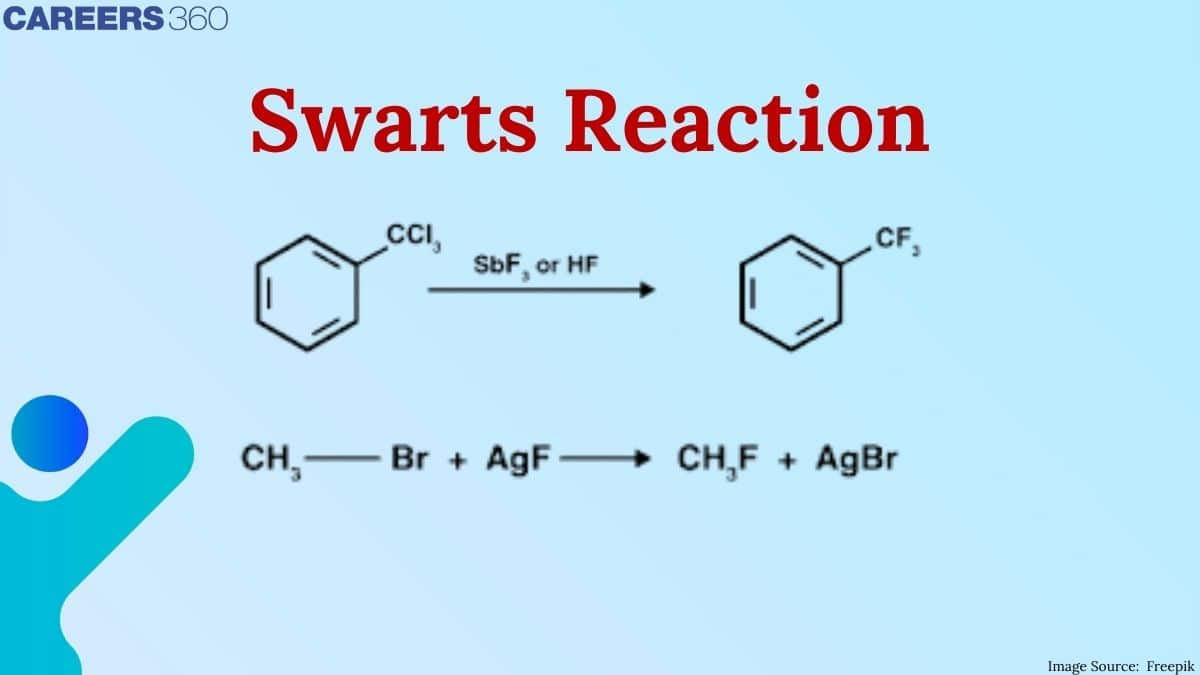
1. $\mathrm{C}_2 \mathrm{H}_5 \mathrm{Cl}+\mathrm{AgF} \rightarrow \mathrm{C}_2 \mathrm{H}_5 \mathrm{~F}+\mathrm{AgCl}$
2. $\mathrm{CH}_3 \mathrm{Br}+\mathrm{AgF} \rightarrow \mathrm{CH}_3 \mathrm{~F}+\mathrm{AgBr}$ ( in the presence of fluoride of heavy metals and swarts reagent)
Swart's reaction is the best reaction for the formation of alkyl fluorides with the help of Swarts reagent, i.e., $\left(\mathrm{SbF}_3+\mathrm{Cl}_2\right)$ and heavy metals fluorides such as $\mathrm{AgF}, \mathrm{F}_2 \mathrm{Hg}_2, \mathrm{CoF}_2$, or $\mathrm{SbF}_3$.
In general, if we have to prepare metal fluorides, we use the Swarts reaction or the Swarts fluorination reaction.
What happens in this reaction?
There will be a small answer based on a concept ( higher electronegative elements replace lower electronegative elements).
According to this concept, we can all well notice that chlorine is generally replaced by fluorine in this reaction. In the presence of antimony trifluoride with antimony salts, the Swart reaction will smoothly move forward due to the +5 oxidation state of the antimony salt.
Swarts Reaction Mechanism
$\text{Step 1: } \text{R}-\underset{\displaystyle|}{\text{X}} + \text{Ag}^+ \rightarrow [\text{R}-\text{X}\cdots\text{Ag}]^+ \text{ (coordination complex)}$
$\text{Step 2: } \text{F}^- \text{ attacks carbon as } \text{Ag}^+ \text{ weakens the C-X bond}$
$\text{R-X} + \text{AgF} \rightarrow \text{R-F} + \text{AgX}\downarrow$
$\text{Driving force: } K_{sp}(\text{AgX}) \ll 1 \Rightarrow \text{AgX precipitates, pushing equilibrium forward}$
$\text{Overall: } \underset{\text{alkyl halide}}{\text{R-X}} + \underset{\text{silver fluoride}}{\text{AgF}} \xrightarrow{\Delta} \underset{\text{alkyl fluoride}}{\text{R-F}} + \underset{\text{silver halide (ppt.)}}{\text{AgX}\downarrow}$
$\text{Where X} = \text{Cl, Br, or I}$
- Swarts' reaction converts alkyl chlorides/bromides/iodides into alkyl fluorides
- The mechanism proceeds via an SN2-like pathway with AgF as the fluorinating agent
- $\mathrm{Ag}^{+}$ acts as a Lewis acid, coordinating with the halogen to weaken the C−X bond
- Precipitation of insoluble $\mathrm{AgX}(\mathrm{AgCl}, \mathrm{AgBr}, \mathrm{AgI})$ is the thermodynamic driving force
- Reactivity order: $\mathrm{R}-\mathrm{I}>\mathrm{R}-\mathrm{Br}>\mathrm{R}-\mathrm{Cl}$
.png)
Applications of the Swarts Reaction
- For the production or formation of alkyl fluoride.
- Used in the formation of freons.
- Swarts ' reaction is used for chemical analysis.
Also read -
Finkelstein Reaction
This is also a halogen exchange method, and its post-reaction product is an alkyl iodide.
Finkelstein reaction occurs through a nucleophilic bimolecular substitution reaction, i.e., SN2 reaction. This is also a type of organic reaction. In this reaction, a primary alkyl halide or pseudohalide reacts with an alkali metal halide to form an alkyl iodide ( this reaction is based on halogen exchange ).
Commonly used reagents in this reaction are:-
- Sodium iodide – acts as a nucleophile in this reaction
- Ethyl chloride – an alkyl halide to complete this reaction.
- The product is Ethyl iodide, i.e., formed due to the exchange between the iodine and chlorine groups. So it is called the halogen exchange process.
Examples of Finkelstein reactions are:-
- $\mathrm{CH}_3 \mathrm{Br}+\mathrm{NaI} \rightarrow \mathrm{CH}_3 \mathrm{I}+\mathrm{NaBr}$ (reaction will happen in the presence of acetone)
- $\mathrm{CH}_3 \mathrm{Cl}+\mathrm{NaI} \rightarrow \mathrm{CH}_3 \mathrm{I}+\mathrm{NaCl}$ (reaction will happen in the presence of acetone)
- $\mathrm{C}_2 \mathrm{H}_5 \mathrm{Cl}+\mathrm{KI} \rightarrow \mathrm{C}_2 \mathrm{H}_5 \mathrm{I}+\mathrm{KCl}$ (this reaction will happen in the presence of acetone)
Related Topics,
Some Solved Examples
Question 1. Which reagents are used in the Swarts reaction?
Ans: The reagents used in Swarts reactions are chlorine or many times antimony pentachloride, also used with antimony trifluoride. The combination of both these reagents (chlorine and antimony trifluoride) is known as a Swarts reagent.
Swarts reagent $=\mathrm{SbF}_3+\mathrm{Cl}_2$
Question 2. What is the product of Finkelstein's reaction?
Ans: Finkelstein's reaction is named after a German chemist. i.e. Hans Finkelstein.
The product of the Finkelstein reaction is an alkyl iodide, which is formed due to halogen exchange. The Finkelstein reaction follows the SN2 ( Nucleophilic Substitution, Second Order ) pathway to form the product.
Question 3. Similarity between Swart's reaction and Finkelstein's reaction?
Ans: Both given reactions are halogen exchange reactions. Both of these reactions follow the SN2 (second-order Nucleophilic substitution) pathway to form a product. In the Swarts reaction, the product is alkyl fluoride, and in the Finkelstein reaction, the product will be alkyl iodide.
Question 4. Difference between the Swarts reaction and the Finkelstein reaction?
Ans: In Swart's reaction, reagents are heavy metal fluorides that can easily fluorinate alkyl components. This reaction is used to form alkyl fluoride with a good yield. The reagents of the Swarts reaction are SbF3 + Cl2, whereas in the Finkelstein reaction, we get alkyl iodide, not fluoride.
The Reactant of this reaction is sodium iodide in acetone ( NaI + C3H6O ). Both of these are organic reactions and play an important role in many organic reactions, analysis, etc.
Question 5: Swarts' reaction is used for the preparation of:
A. Fluoroalkanes
B. Chloroalkanes
C. Bromoalkanes
D. Iodoalkanes
Solution: Swarts' reaction involves the replacement of halogen (usually $\mathrm{Cl} / \mathrm{Br}$ ) by fluorine using metal fluorides like $\mathrm{AgF}_1 \mathrm{SbF}_3 \rightarrow$ formation of fluoroalkanes.
Hence, the correct answer is option (A)
Question 6: In Swart's reaction, the halide most commonly replaced is:
A. F
B. Cl
C. Br
D. I
Solution: Chloroalkanes are generally converted into fluoroalkanes using metal fluorides.
Hence, the correct answer is option (B)
Question 7. The Finkelstein reaction is mainly carried out in:
A. Aqueous medium
B. Acetone
C. Benzene
D. Water–ethanol mixture
Solution: Finkelstein reaction: $\mathrm{R}-\mathrm{Cl} / \mathrm{R}-\mathrm{Br}+\mathrm{NaI} \rightarrow \mathrm{R}-\mathrm{I}+\mathrm{NaCl} / \mathrm{NaBr}$ (in acetone).
$\mathrm{NaCl} / \mathrm{NaBr}$ precipitates out → reaction driven forward.
Hence, the correct answer is option (B)
Question 8. Is F2Hg2 a reagent of the Swarts reaction?
Ans: Yes, it is a very good fluorinating agent and also a reagent for Swarts reaction.
$\mathrm{F}_2 \mathrm{Hg}_2$ – Compound name is “ Mercury(I) fluoride “
Question 9. What is Swart's reaction?
Ans: It is a process by which chlorine-containing organic compounds are changed to fluorides through a halogen exchange process with the help of Swarts reagent, i.e., in the presence of antimony trioxide and chlorine. This reaction follows the SN2, i.e., Nucleophilic substitution pathway.
Also, check-
Frequently Asked Questions (FAQs)
- Poor reactivity with sterically hindered or tertiary centers – elimination dominates.
- No direct fluorination of aryl C–Cl/C–Br bonds – other methods required.
- Moisture sensitivity – water deactivates SbF₃ and creates HF, potentially corroding equipment.
- Toxic antimony waste – must be managed and can increase the cost of large‑scale processes.
- Limited functional‑group tolerance – acids, strong nucleophiles, and bases can interfere.
The Swarts reaction is a halogen‑exchange (halogenation) process that converts alkyl or alkenyl chlorides/bromides into the corresponding fluorides.
It is typically carried out with antimony(III) fluoride (SbF₃) or antimony(V) fluoride (SbF₅) in the presence of a halogenated substrate and a Lewis base (often pyridine or an amine). The reaction was first reported by the Belgian chemist Frédéric Swarts in 1896.

