Grignard Reagent - Structure, Preparation, Application, Uses, FAQs
Have you ever wondered how a whole molecule is formed just by starting with something as simple as an alkyl halide? The answer to this question will be known after reading this article on the Grignard reagent. Grignard reagent is an Organometallic compound written as RMgX, where R is an alkyl or aryl group and X is a halogen. Grignard reagents are highly reactive organometallic compounds that contain carbon–magnesium bonds and play an important role in the formation of carbon–carbon bonds.
This Story also Contains
- Structure Of Grignard Reagent
- Preparation Of Grignard Reagent
- Applications Of Grignard Reagents
- Uses Of Grignard Reagent
- Some Solved Examples
A French Chemist named Francois Grignard discovered this reagent in 1900. He was investigating the organic reactions of halides with metals, and while reacting different metals with organic halides, he noticed that the reaction of magnesium with organic halides in the presence of dry ether forms a very reactive organo-magnesium compound. Due to this discovery, he was awarded the Nobel Prize in 1912. In this article, we will cover the topic of the Grignard Reagent. This topic falls under the broader category of Haloalkanes And Haloarenes, which is a crucial chapter in (Class 12 Chemistry). It is not only essential for board exams but also for competitive exams like the JEE Mains Exam , National Eligibility Entrance Test (NEET), and other entrance exams such as SRMJEE, BITSAT, WBJEE, and more.
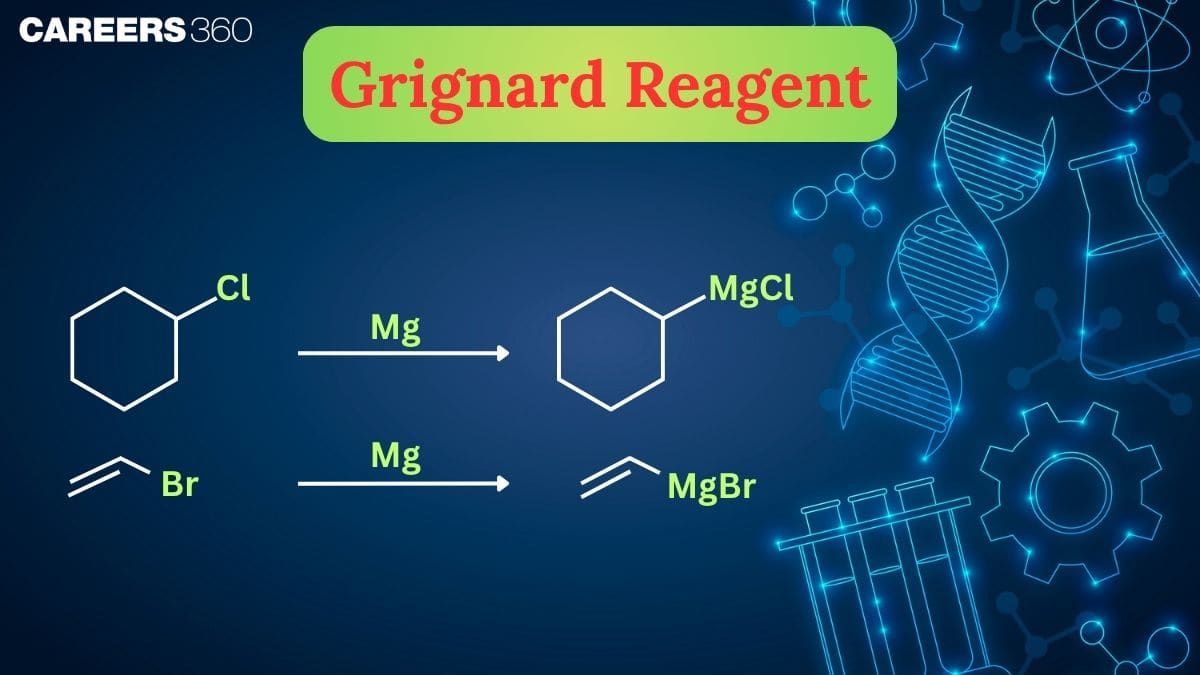
Structure Of Grignard Reagent
A Grignard reagent is an organometallic compound having the general formula: RMgX
where:
- R = Alkyl or aryl group
- Mg = Magnesium atom
- X = Halogen atom such as chlorine (Cl), bromine (Br), or iodine (I)
Structural Features
- In a Grignard reagent, carbon is directly bonded to magnesium.
- The carbon–magnesium bond $(\mathrm{C}-\mathrm{Mg})$ is highly polar because magnesium is more electropositive than carbon.
- Carbon acquires a partial negative charge, while magnesium gets a partial positive charge.
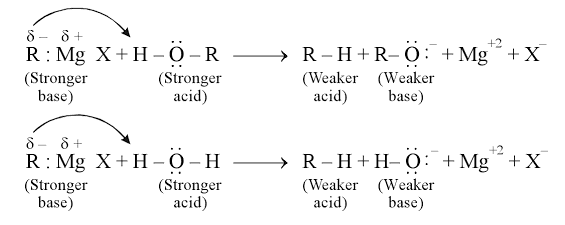
- Due to this polarity, Grignard reagents behave as strong nucleophiles and strong bases.
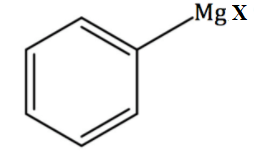
Preparation Of Grignard Reagent
Grignard reagents are prepared by reacting magnesium metal with an organic halide in the presence of dry ether. These organomagnesium compounds are highly reactive and require strictly anhydrous conditions for their preparation and storage.
$\mathrm{RX}+\mathrm{Mg} \xrightarrow[\text { dry ether }]{ } \mathrm{RMgX}$
where:
- R = Alkyl or aryl group
- X = Halogen atom such as chlorine, bromine, or iodine
Explanation
- Magnesium inserts itself between the carbon atom and the halogen atom of the organic halide to form the Grignard reagent.
- Ether solvents such as diethyl ether are used because they stabilize the organomagnesium compound through coordination with magnesium.
- The reaction must be carried out under air-free and moisture-free conditions because Grignard reagents react rapidly with water, oxygen, and other protic substances.
Importance of Anhydrous Conditions
Grignard reagents undergo protonolysis in the presence of water or alcohols, which destroys the reagent.
$\mathrm{RMgX}+\mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{RH}+\mathrm{Mg}(\mathrm{OH}) \mathrm{X}$
Therefore:
- All apparatus must be completely dry.
- Moisture and air must be avoided during the preparation.
However, this compound can be formed in solution by the use of ultrasound since it activates the magnesium and, thereby, consumes water present in the solution. An anhydrous condition is more suitable for the preparation of the Grignard reagent. Following the correct procedures results in the formation of the Grignard reagent. The following reaction shows the preparation of the Grignard reagent from an organic halide.
Applications Of Grignard Reagents
1. Formation of Alkane
Grignard reagent, on reaction with any compound containing an active H atom, produces an alkane.
$\mathrm{MgRX}+\mathrm{HZ} \rightarrow \mathrm{MgXZ}+\mathrm{RH}$
2. Formation Of Alkene
Unsaturated halide with Grignard reagent gives alkene
$\mathrm{CH}_3 \mathrm{MgI}+\mathrm{CH}_2=\mathrm{CHI} \longrightarrow \mathrm{CH}_2=\mathrm{CHCH}_3+\mathrm{MgI}_2$
3. Formation of Alkynes
Lower alkyne $+G R \rightarrow$ Product $\xrightarrow[\text { haldide }]{\text { alks }}$ Higher alkyne
$\mathrm{R}-\mathrm{C} \equiv \mathrm{C}-\mathrm{H}+\mathrm{CH}_3 \mathrm{MgBr} \xrightarrow[-\mathrm{CH}_4]{ } \mathrm{R}-\mathrm{C} \equiv \mathrm{C}-\mathrm{MgBr}$
4. Formation of Alcohol
(a) Primary alcohol: Obtained from :
(i) Dry oxygen
(ii) Epoxy ethane
(iii) Formaldehyde
(b)Secondary Alcohol: obtained from:
(i) all aldehydes except formaldehyde
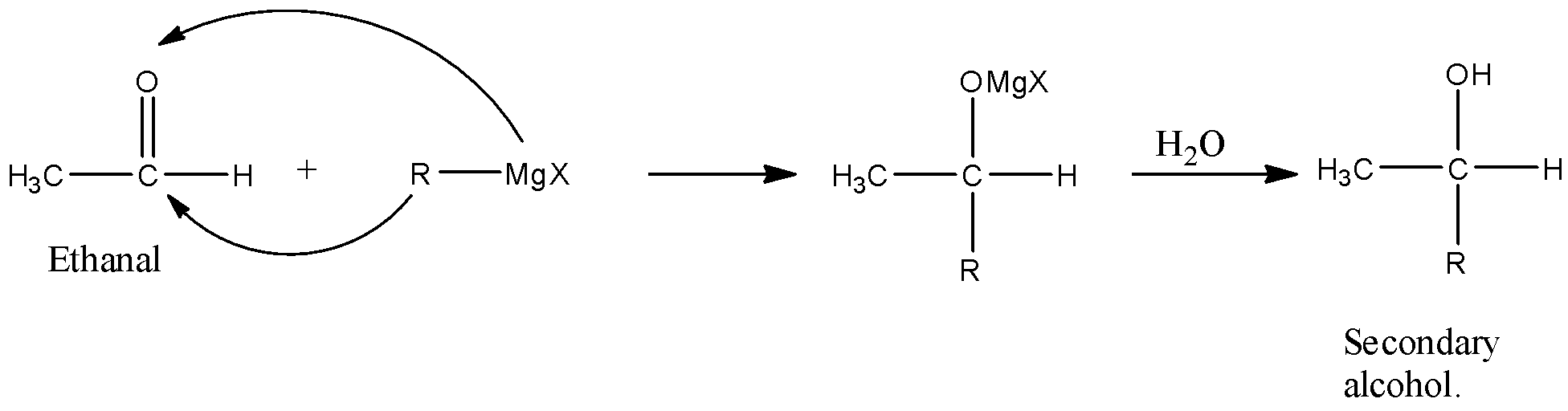
(ii) Ethyl formate + 2 moles of RMgX
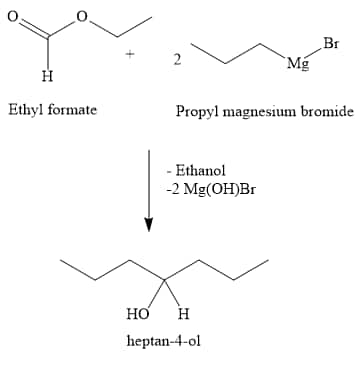
(c) Tertiary Alcohol: Obtained from:
(i) All esters except ethyl formate
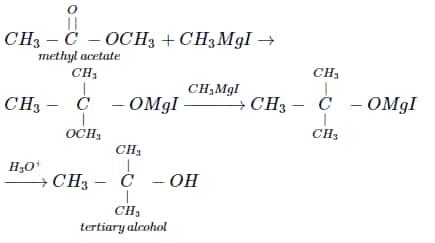
(ii) All ketones
.png)
(iii) (RCOCl) + 2 moles of RMgX
.png)
5. Formation of Aldehydes:
(i) From HCN
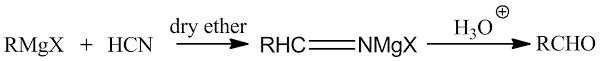
(ii) From ethyl formate + 1 mole of RMgX
.png)
6. Formation of ketones:
(i) Alkyl Cyanide
$\mathrm{CH}_3 \mathrm{CN}+\mathrm{C}_2 \mathrm{H}_5 \mathrm{MgBr} \rightarrow \mathrm{CH}_3 \mathrm{COC}_2 \mathrm{H}_5+\mathrm{NH}_3+\mathrm{MgBrOH}$
(ii) Acetyl Chloride
$\mathrm{CH}_3 \mathrm{COCl}+\mathrm{CH}_3 \mathrm{MgX} \rightarrow \mathrm{CH}_3 \mathrm{COCH}_3+\mathrm{MgXCl}$
Reaction With Carbonyl Compounds
Grignard reagents react with carbonyl compounds like ketones and aldehydes to form corresponding alcohols. The nature of the substituent that gets attached to the carbonyl compound determines the product. When methanal is used as an aldehyde, the obtained alcohol will be primary, and if any aldehyde other than this is used, a secondary aldehyde is obtained. It can also be used for the alkylation of aldehydes and ketones. The Grignard reagent acts as a nucleophile, and thereby nucleophilic substitution reactions take place. The figure below shows the reaction of the Grignard reagent to form Benzyl alcohol.
Uses Of Grignard Reagent
- Grignard reagents are a very important organic compound that has many applications in the chemical field, mainly the organic chemistry field.
- Grignard reagents have industrial applications, also. The very important use of the Grignard reagent is the production of tamoxifen a medicine used for the treatment of breast cancer. The reaction below shows the preparation of tamoxifen.
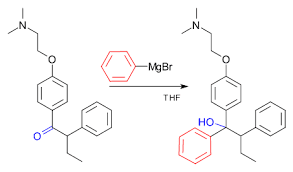
- Grignard reagents can be used to produce alcohol from epoxides.
- Grignard reagent reaction with aldehyde, ketone, and esters to form alcoholic compounds.
- It can be used for the degradation reaction of Grignard reagents used for the chemical analysis of triacylglycerol, and also for some cross-coupling reactions that are involved in the formation of carbon-carbon and carbon-hydrogen bonds.
- It can be used for the synthesis of many organometallic compounds.
- Grignard reagents are best for the preparation of amides, acetals, amino compounds, organosulfur compounds, ethers, ketones, aldehydes, etc.
- It can be used for the production of several compounds that have a very important application in the pharmaceutical, perfume, and specialty chemicals fields.
- Grignard reagents are used for the production of optically active compounds by the reaction of a secondary alcohol with the alkyl halides in the presence of a chiral phosphine metal as a catalyst.
Also check-
Some Solved Examples
Question 1: When phenyl magnesium bromide reacts with tert. Butanol, which of the following is formed?
1)Tert. butyl methyl ether
2) (correct)Benzene
3)Tert. butyl benzene
4)Phenol
Solution:
As we learned,
Zerewitinoff Method -
The reaction of the alcohol with the Grignard reagent.
$R^{\prime} OH+RMgX \rightarrow RH+R^{\prime} OMgX$
$\mathrm{C}_6 \mathrm{H}_5 \mathrm{MgBr}+\left(\mathrm{CH}_3\right)_3 \mathrm{C}-\mathrm{OH} \rightarrow \mathrm{C}_6 \mathrm{H}_6+\left[\left(\mathrm{CH}_3\right)_3 \mathrm{Co}\right] \mathrm{MgBr}$
Hence, the answer is option (2).
Question 2: Reaction of ROH with R'MgX produces:
1) RH
2) (correct)R'H
3) R-R
4) R-R'
Solution:
Alkyl magnesium halides(RMgX) are called Grignard reagents. These undergo double decomposition reactions with water, ammonia, alcohol or amine having active H atom(attached to strongly electronegative O, N, S, or F and triple bond, etc.) to give alkane corresponding to an alkyl group of Grignard reagent. The reaction occurs as follows:

So,
$\mathrm{R}^{\prime} \mathrm{MgX}+\mathrm{ROH} \rightarrow \mathrm{R}^{\prime} \mathrm{H}+\mathrm{Mg}(\mathrm{OR}) \mathrm{X}$
Hence, the answer is option (2)
Question.3 Which of the following compounds will form a hydrocarbon on reaction with a Grignard reagent?
1) (correct) $\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{OH}$
2) $\mathrm{CH}_3 \mathrm{CHO}$
3) $\mathrm{CH}_3 \mathrm{COCH}_3$
4) $\mathrm{CH}_3 \mathrm{CO}_2 \mathrm{CH}_3$
Solution:
Reaction of Grignard reagent with H2O -
Alkane / Benzene is obtained
- wherein
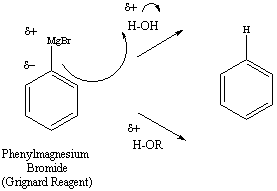
Reaction of Grignard reagent with Alcohol -
Alkane is obtained.
Hence, the correct answer is option (1)
Question 4: What is the major product formed when methyl magnesium bromide reacts with formaldehyde followed by hydrolysis?
$\mathrm{CH}_3 \mathrm{MgBr}+\mathrm{HCHO} \xrightarrow[\mathrm{H}_3 \mathrm{O}^{+}]{ } \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{OH}$
(A) Methanol
(B) Ethanol (Correct)
(C) Propanol
(D) Acetaldehyde
Solution:
Grignard reagents react with formaldehyde to form primary alcohols after hydrolysis. Here, methyl magnesium bromide gives ethanol.
Hence, the correct answer is option (B)
Question 5: Which of the following compounds will destroy a Grignard reagent immediately?
(A) Dry ether
(B) Benzene
(C) Water (Correct)
(D) Toluene
Solution:
Grignard reagents are highly reactive and react rapidly with water to form hydrocarbons.
$\mathrm{RMgX}+\mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{RH}+\mathrm{Mg}(\mathrm{OH}) X$
Therefore, Grignard reagents must always be prepared and used under anhydrous conditions.
Hence, the correct answer is option (C)
Practice More Questions From The Link Given Below
For more questions to practice, the following MCQs will help in the preparation for competitive examinations
Some related topics:
Frequently Asked Questions (FAQs)
An organomagnesium compound with the general formula RMgX is known as a Grignard Reagent. Where R represents organic groups such as alkyl or aryl, X represents Halogen gases, and Mg is the symbol of Magnesium.
Reaction of organic halide with magnesium in anhydrous ether leads to the formation of Grignard reagent. It is essential for the reaction to conduct in anhydrous condition because Grignard reagents react vigorously with water.
The Grignard reagent is prepared under anhydrous conditions is because of the reaction of the Grignard reagent with water. It reacts very quickly with any proton-containing compound and forms a hydrocarbon. So the effect of the Grignard reagent and its application is lost. The removal of moisture before conducting the preparation is very necessary.
R-Mg-X+H2O→R-H+Mg(OH)X
The Grignard reagent is highly reactive. There are certain precautions to consider while handling Grignard reagent:
- Must be kept away from water.
- Gloves and Goggles should be used while handling
- Reactions of the Grignard reagent must be carried out in a ventilated place
- Proper disposal
Yes, Grignard reagent reacts with many solvents, particularly those containing acidic protons such as alcohol, water, and acids.
Grignard reagents are widely used in organic chemistry :
- In the formation of alcohols by nucleophilic addition to carbonyl compounds (aldehydes and ketones).
- In the synthesis of various complex organic molecules.
- The preparation of carboxylic acids and other functional groups via reactions with carbon dioxide.
When a Grignard reagent reacts with water, it forms an alkane and magnesium hydroxide. For example:
R-Mg-X + H2O→ R-H + Mg(OH)X

