Wave Number - Definition, Formula, Symbol, FAQs
Wavenumber is a simple concept used in physics and chemistry to describe how many waves are present in a given distance. It helps us understand how tightly waves are packed together. The smaller the wavelength, the higher the wavenumber, which means more waves in less space. It is commonly used in topics like light, sound, and spectroscopy. In spectroscopy, wavenumber is usually written in cm⁻¹, making calculations easier while studying energy and molecular vibrations. Learning wavenumber, its meaning, and its uses makes it easier to understand wave behavior, energy changes, and important concepts in modern physics and chemistry.
This Story also Contains
- What is Wave Number?
- Wavenumber Definition
- Dimensional Formula of Wavenumber (k)
- Applications of Wavenumber
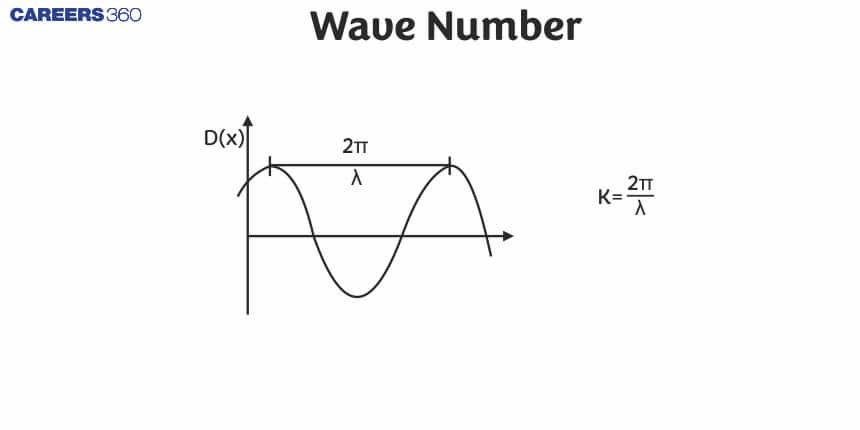
What is Wave Number?
Wavenumber is defined as the number of complete wave cycles present in a unit distance. It is an important concept in wave mechanics, optics, and physics.
The wavenumber equation is given by:
$k=\frac{2 \pi}{\lambda}$
- $k$ is the wavenumber (also called angular wavenumber)
- $\lambda$ is the wavelength of the wave
Unit: The SI unit of wavenumber is rad/m (radian per meter).
Another commonly used form is spectroscopic wavenumber:
$\tilde{\nu}=\frac{1}{\lambda}$
which is measured in $\mathbf{m}^{\mathbf{- 1}}$ or $\mathbf{c m}^{\mathbf{- 1}}$.
Also Read:
Wavenumber Definition
Wavenumber is a fundamental quantity used in physics, chemistry, and spectroscopy to describe wave properties in terms of spatial variation.
1. In Theoretical Physics
Wavenumber is defined as the number of radians per unit distance. It represents how rapidly the phase of a wave changes in space.
2. In Chemistry and Spectroscopy
Wavenumber is defined as the number of waves per unit distance. It is commonly used in spectroscopy and is expressed as:
$\bar{\nu}=\frac{1}{\lambda}$
Unit: $\mathbf{c m}^{\boldsymbol{-} \mathbf{1}}$ (commonly used in spectroscopy)
3. Complex Wavenumber
In a medium, the wavenumber can be written as:
$
k=k_0 \sqrt{\varepsilon \mu}=k_0 n
$
Where:
- $k_0=$ free-space wavenumber
- $\varepsilon=$ relative permittivity
- $\mu=$ relative permeability
- $n=$ refractive index
The imaginary part of wavenumber represents attenuation, which is important in studying exponentially decaying (evanescent) waves.
Wavenumber Equation
The angular wavenumber is given by:
$
k=\frac{2 \pi}{\lambda}=\frac{\omega}{v_p}
$
Where:
- $v_p=$ phase velocity
- $\omega=2 \pi \nu=$ angular frequency
Also read -
Dimensional Formula of Wavenumber (k)
Wavenumber represents the number of waves per unit length. Since it is the reciprocal of wavelength, its dimensional formula is:
$
[k]=\left[M^0 L^{-1} T^0\right]
$
- No dependence on mass or time
- Depends only on length (inverse of length)
Applications of Wavenumber
Wavenumber is widely used in physics and chemistry to describe wave properties in a simple form.
- Spectroscopy: Used to represent spectral lines in $\mathbf{c m}^{-\mathbf{1}}$ for easy analysis of molecular and atomic transitions.
- Energy Relation: Directly related to energy of radiation $(E \propto \bar{\nu})$, useful in quantum studies.
- Wave Mechanics: Helps in expressing wave equations and phase variation using $k=\frac{2 \pi}{\lambda}$.
- Optics: Used to study refraction and dispersion as wavenumber changes with medium ( $k=k_0 n$ ).
- Quantum Mechanics: Represents particle momentum ( $p=\hbar k$ ).
Also check-
- NCERT Exemplar Class 11th Physics Solutions
- NCERT Exemplar Class 12th Physics Solutions
- NCERT Exemplar Solutions for All Subjects
NCERT Physics Notes:
Frequently Asked Questions (FAQs)
Wave vector, symbol k, is a property of wave which shows the direction and magnitude of wave number or else angular wave number.
It is considered as phase lag per unit distance.
It is significant in both the fields of physics and chemistry.
We see that wave number has vast use in communication studies and also in quantum mechanics. Whereas in chemistry it plays important role for studying spectroscopy.
Nu is the symbol of frequency used in science. Frequency is also a property of electromagnetic wave and is defined as number of complete cycles per second. Hence its SI unit is HERTZ. Dimensional formula is given as[M0L-1T0]OR T-1. In Sound energy, Frequency denotes the pitch of any wave/sound. That is, how much shrill or sharp the voice is.
Wavelength is also a very important component of any electromagnetic wave. Wavelength is the distance between the two successive crests or troughs. Angular width is used in diffraction phenomenon and is known as the fringe width β denoted by the symbol beta. Its expression is given as
β= λD/d, where λ is the wavelength of source, D is the distance between two source and the screen and d is the distance between the two slits.
Wave number is a scalar quantity whereas wave vector is a vector quantity representing the magnitude and direction of propagation of electromagnetic waves.
Waves created due to the disturbance in electromagnetic field is known as EM waves. It consists of seven different types of waves. It travels with the velocity of 3 x 108 m/s in vacuum space. Its properties are wavelength, frequency, phase difference, amplitude and intensity. In today’s era there is vast use of electromagnetic waves. Studying and working in this field has in turn changed the scenario of this technology progressive generation.
No, in sound waves loudness of voice is associated with the higher amplitude. Higher frequency means the pitch of the voice is high. Pitch signifies the shrillness and the sharpness of the sound.

