Nitrogen Fixation: Definition, Process, Examples, Types, & Facts
Nitrogen fixation is the biological and chemical conversion of inert atmospheric nitrogen (N₂) into ammonia (NH₃) or related compounds usable by plants. This process, driven by nitrogen-fixing bacteria and the nitrogenase enzyme, forms the foundation of the nitrogen cycle essential for life on Earth.
This Story also Contains
- Importance Of Nitrogen
- The Nitrogen Cycle
- What Is Nitrogen Fixation?
- Mechanism Of Biological Nitrogen Fixation
- Types of Nitrogen-Fixing Bacteria
- Role of Legumes in Nitrogen Fixation
- Factors Affecting Nitrogen Fixation
- Applications Of Nitrogen Fixation
- Nitrogen Fixation NEET MCQs (With Answers & Explanations)
- Recommended video on Nitrogen Fixation

Importance Of Nitrogen
Nitrogen is a basic constituent of the makeup of all life on Earth. It is found in amino acids, proteins, enzymes, tissues and hormones. Nitrogen comprises about 78% of atmospheric gases; however, it needs to be fixed into a form usable by plants. This is a step necessary for the synthesis of proteins, DNA, and other important compounds.
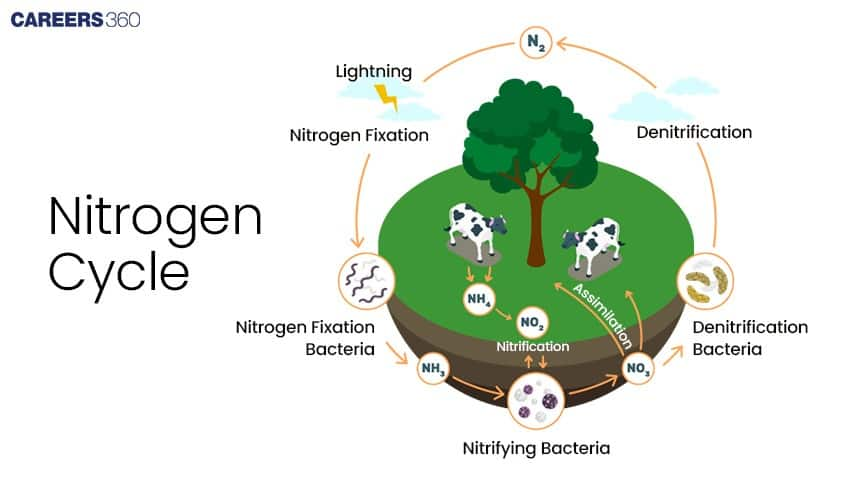
The Nitrogen Cycle
The nitrogen cycle is the biogeochemical process by which nitrogen is changed into its various forms as it circulates through an ecosystem. This cycle maintains the nitrogen balance in the environment. The important stages of the nitrogen cycle involve:
Nitrogen Fixation: The atmospheric nitrogen, N₂, is converted to ammonia, NH₃.
Nitrification: The ammonia is transformed first into nitrites, NO₂, and then nitrates, NO₃.
Nitrate Assimilation: Plants absorb nitrates to synthesize proteins.
Ammonification: Organic nitrogen gets decomposed into ammonia.
Denitrification: Nitrates are reduced back to atmospheric nitrogen in the air, completing the cycle.
What Is Nitrogen Fixation?
Nitrogen fixation is a chemical process of reformatting inert atmospheric nitrogen either into ammonia or related nitrogenous compounds, making them available to most living organisms. This process is important since nitrogen forms the basis for amino acids, proteins, and nucleic acids. While nitrogen makes up a huge part of the Earth's atmosphere, it needs to be changed into forms usable by plants and, indirectly, the animals that graze on them.
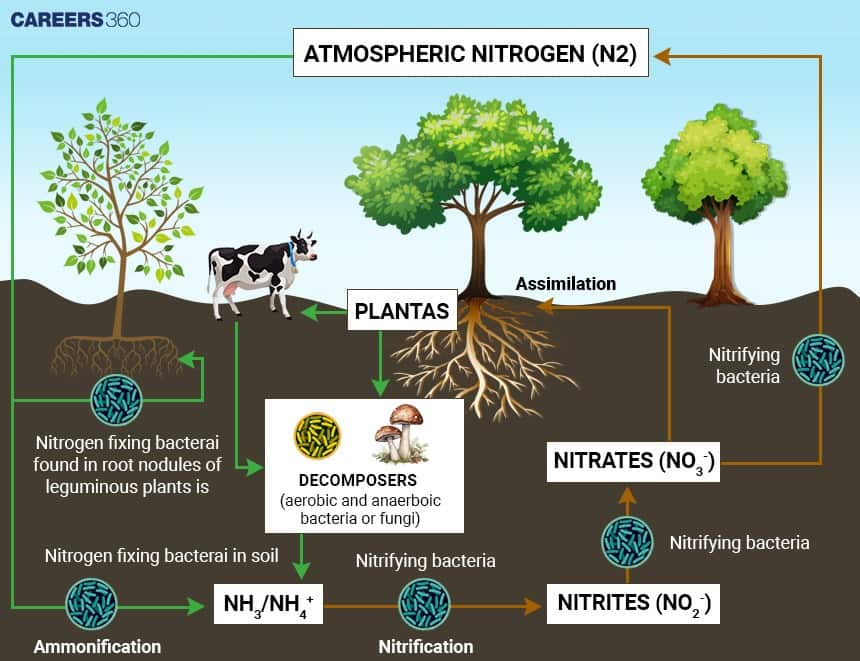
Mechanism Of Biological Nitrogen Fixation
Biological nitrogen fixation is done mainly by a few bacteria having the enzyme nitrogenase. This enzyme catalyzes the conversion of nitrogen gas to ammonia according to the following equation:
N2 + 8H++ 8e− + 16ATP → 2NH3 + H2 + 16ADP + 16Pi
In this reaction, atmospheric nitrogen reacts with protons and electrons in the presence of ATP, which generates ammonia.
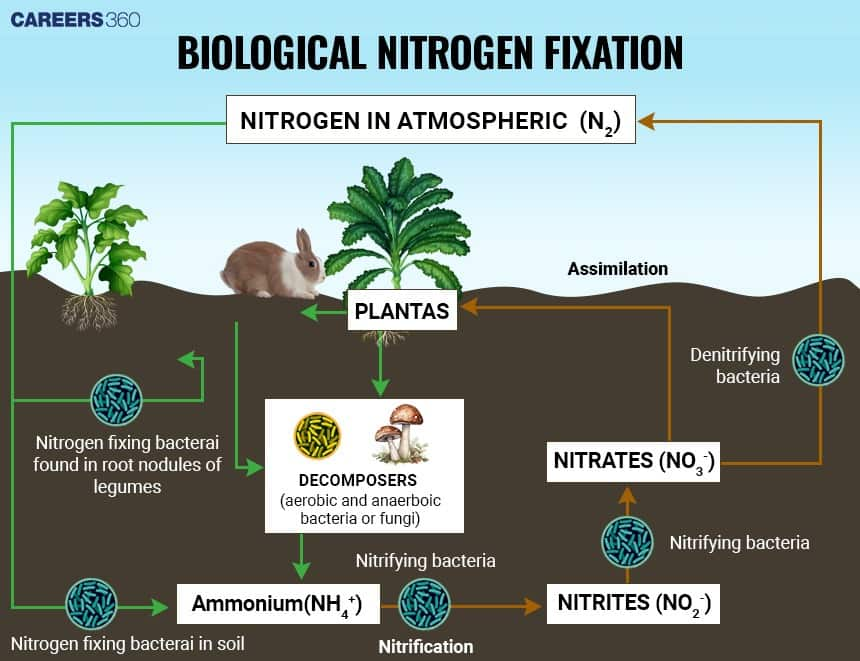
Types of Nitrogen-Fixing Bacteria
There are two major divisions regarding nitrogen-fixing bacteria:
Symbiotic Nitrogen-Fixing Bacteria
These bacteria are known to form symbiotic relationships with certain plants, especially legumes. Probably the most well-known example is Rhizobium, which infects the root hairs of leguminous plants and induces the formation of root nodules. Inside these nodules, the bacteria fix atmospheric nitrogen into ammonia, which is used by the plant for growth. In turn, the plant provides the bacteria with carbohydrates and a safe environment.
Non-Symbiotic Nitrogen-Fixing Bacteria
These bacteria do not associate with the plant and live freely in the soil. Examples include Azotobacter and Clostridium. They also fix atmospheric nitrogen and increase the amount of nitrogen available to plants in the soil.
Associative Nitrogen-Fixing Bacteria
These nitrogen fixing bacteria live in close association with the roots but they do not enter in the plant tissues. These bacteria, for example Azospirillum, colonise roots, fix atmospheric nitrogen and receive nutrients from the plants. This relationship is also called associative symbiosis.
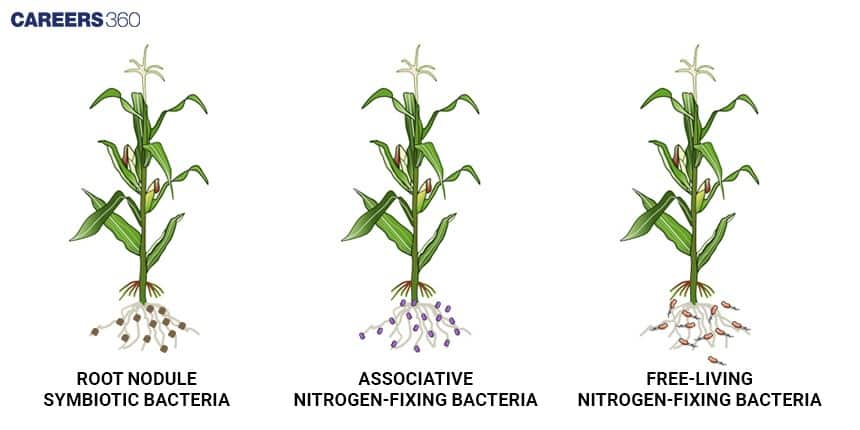
Role of Legumes in Nitrogen Fixation
Leguminous plants have a symbiotic relationship with nitrogen-fixing bacteria. Hence, they are involved in nitrogen fixation. The formation of nodules on the roots allows for effective nitrogen uptake, hence the reason why legumes are included in crop rotation and sustainable agriculture. When legumes are planted, it enhances the natural amount of nitrogen in the soil, which means farmers do not need synthetic fertilizer to raise plants.
Factors Affecting Nitrogen Fixation
Several factors will determine how effective nitrogen fixation will be. Some of the factors include:
Factors | Effect |
Soil pH | pH should be within ideal levels, usually between 6.0 and 7.5, to ensure optimum activity of the bacteria. |
Moisture Levels | Adequate moisture to help the bacteria thrive and the biochemical process ensues accordingly. |
Temperature | Nitrogen-fixing bacteria need specific temperatures to attain their optimum activity. |
Nutrient Availability | Availability of other nutrients can impact the whole health status of the plant or its ability to support nitrogen-fixing bacteria. |
Oxygen Level | Nitrogenase is oxygen sensitive bacteria, leghemoglobin regulates the supply of oxygen in the root nodules |
Applications Of Nitrogen Fixation
The applications of nitrogen fixation includes:
Biofertilizers: Using bacteria like Rhizobium, Azospirillum and Anabaena in sustainable farming.
Environmental Balance: Maintains the levels of nitrogen in the ecosystems.
Agricultural Productivity: Reduces the use of chemical fertilizers.
Soil Reclamation: Enhances the nitrogen levels in the soil and makes it suitable for growing crops.
Nitrogen Fixation NEET MCQs (With Answers & Explanations)
The key concepts to be covered under this topic for different exams are:
Steps of the Nitrogen cycle
Types of Nitrogen fixing bacteria
Factors affecting Nitrogen fixation
Practice Questions for NEET
Q1. Which of the following organisms does not fix nitrogen?
Azotobacter
Nostoc
Spirogyra
Anabaena
Correct answer: 3) Spirogyra
Explanation:
Spirogyra is a genus of green algae that is not capable of fixing atmospheric nitrogen. Other than some cyanobacteria, other algae, for example, have the necessary enzyme, known as nitrogenase and the nif gene for fixation of nitrogen, Spirogyra lacks such. This places it on the disadvantageous side when it comes to contribution to the provision of nitrogen to an aquatic ecosystem, meaning it gets most of its nitrogen from external sources instead of fixing atmospheric nitrogen.
Hence, the correct answer is option 3) Spirogyra.
Q2. Minerals involved in carbohydrate translocation, maintaining ribosome structure and activation of nitrogenase respectively are
Mn, B, Ca
Ca, Mg, Mo
B, Mn, Mo
Cu, mg, B
Correct answer: 3) B, Mn, Mo
Explanation:
Minerals involved in carbohydrate translocation, maintaining ribosome structure, and activation of nitrogenase respectively are boron, manganese, and molybdenum.
Hence, the correct answer is option 3) B, Mn, Mo.
Q3. The common nitrogen-fixer in paddy fields is
Frankia
Rhizobium
Azospirillum
Oscillatoria
Correct answer: 3) Azospirillum
Explanation:
Plants cannot use nitrogen from the atmosphere. There are nitrogen-fixing bacteria that convert atmospheric nitrogen into ammonium compounds and supply them to plants. Examples include Azotobacter, Frankia, Rhizobium, and Azospirillum. It can be seen that Azospirillum is widely used in rice fields as a nitrogen-fixing organism. It is used as a bio-fertilizer in rice fields. It promotes plant growth. So the correct answer is "Azospirillum".
Hence, the correct answer is option 3) Azospirillum.
Also Read:
Recommended video on Nitrogen Fixation
Frequently Asked Questions (FAQs)
Nitrogen is needed for amino acids, proteins, and nucleic acids, and therefore for all growth and developmental processes of plants.
Nitrogen fixation is the process of taking atmospheric nitrogen, N₂ and reducing it to ammonia or related nitrogenous compounds that the plant can use.
The legume associates symbiotically with nitrogen-fixing bacteria, which enables the plants to convert atmospheric nitrogen into ammonia for their growth and to enhance the nitrogen conditions of the soil.
These include soil pH, soil moisture, temperature, and other nutrients that are all implicated in influencing the activity of nitrogen-fixing bacteria.

