Classification of Colloids - Types of Colloids, Definition, FAQs
Have you ever wondered how substances like milk, ink, or even fog form? Looking at them, it doesn't seem like they are a mixture of two substances - but they actually are. These are examples of colloids, where particles are dispersed within another substance but are not large enough to settle out or be filtered. Colloids are a type of mixture in which one substance (called the dispersed phase) is finely distributed throughout another substance (called the dispersion medium). Colloidal particles are intermediate in size, larger than molecules in a solution but smaller than particles in a suspension.
This Story also Contains
- What Is a Colloid?
- Dispersion Medium And Dispersed Phase
- Classification Of Colloids
- Classification Of Colloids Based on Physical State
- Classification Of Colloids Based on Interaction Between Phases
- Types of Colloids Based on Type of Particles
- Classification of Colloids Based on the Interaction Medium
- Some Solved Examples
In this article, we will discuss the classification of colloids, the dispersion phase, and the dispersion medium, types of colloids, and some solved examples related to them. To know more about colloids, scroll down.
What Is a Colloid?
A colloid is a heterogeneous mixture in which minute particles of one component are scattered in a dispersion medium of another substance.
The tiny particles in this combination range in size from 1 to 1000 nanometres in diameter, but they remain suspended and do not settle to the bottom. They can be seen with an optical or electron microscope (smaller particles).
A dispersed phase and a dispersion medium make up colloids. They're divided into groups based on the features of the dispersed phase and the medium. Let us discover more about them right now.
The following is the IUPAC definition of colloid:
“A colloidal state is a condition of dispersion in which molecules or polymeric particles with at least one dimension between 1 nanometre and 1 micrometre are distributed in a medium.”
Dispersion Medium And Dispersed Phase
Dispersed Phase
The dispersed phase is the substance that is present in the form of small particles and is distributed throughout the other substance.
- It is similar to the solute in a true solution.
- The particles of the dispersed phase have sizes between 1 nm and 1000 nm.
Examples
- In milk, fat globules are the dispersed phase.
- In fog, water droplets are the dispersed phase.
Dispersion Medium
The dispersion medium is the substance in which the dispersed phase is distributed.
- It is similar to the solvent in a true solution.
- It forms the continuous phase of the colloidal system.
Examples
- In milk, water is the dispersion medium.
- In smoke, air is the dispersion medium.
Also Read:
Classification Of Colloids
Colloids are classified on the basis of:
- Physical state of dispersed phase and dispersion medium
- Nature of interaction between dispersed phase and dispersion medium
- Type of particles of the dispersed phase
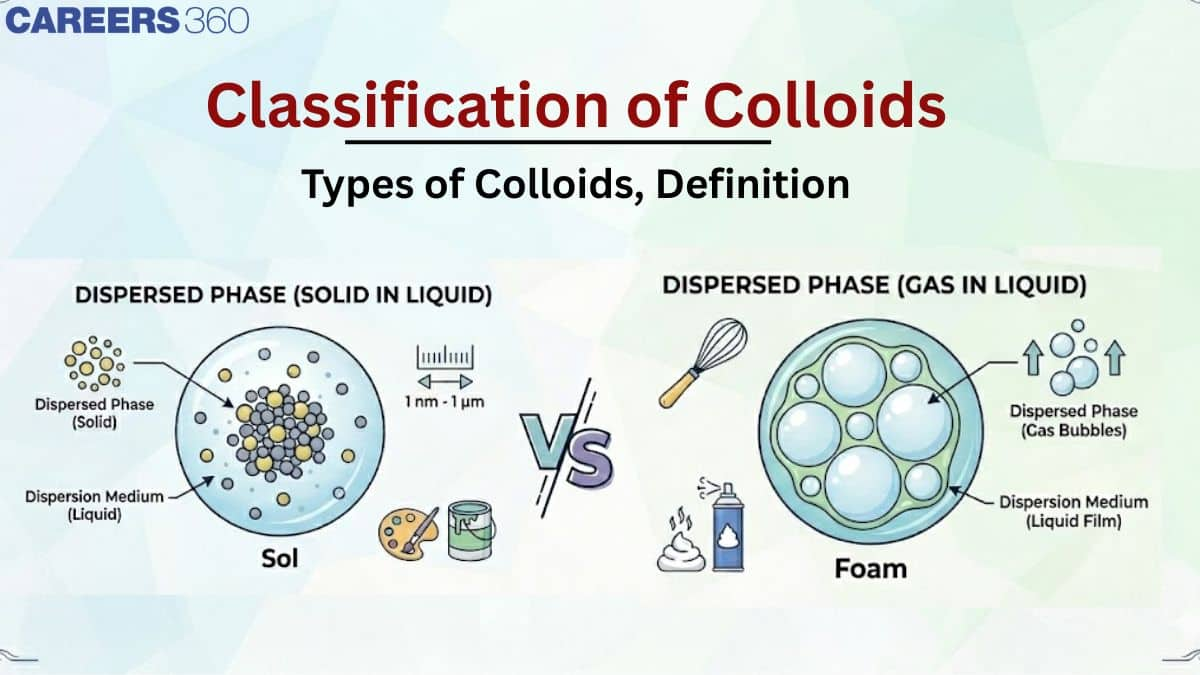
Classification Of Colloids Based on Physical State
Colloids are classified based on the nature of the interaction between the dispersed phase and the dispersion medium.
| Dispersed Phase | Dispersion Medium | Type of Colloid | Example |
|---|---|---|---|
| Solid | Solid | Solid Sol | Colored glass, Marbles |
| Solid | Liquid | Sol | Paint, mud |
| Solid | Gas | Aerosol | Smoke |
| Liquid | Solid | Gel | Jelly, cheese |
| Liquid | Liquid | Emulsion | Milk |
| Liquid | Gas | Aerosol | Fog, clouds |
| Gas | Solid | Solid Foam | Pumice stone, Cake |
| Gas | Liquid | Foam | Shaving cream |
Classification Of Colloids Based on Interaction Between Phases
Lyophilic Colloids
A lyophilic colloid is one in which the dispersed phase has an affinity for the dispersion medium. The words lyo and philic, respectively, signify "liquid" and "loving." Even if the dispersed phase and the dispersion medium are separated, they can easily be reconstituted by simply mixing them together. Furthermore, because of their sturdy nature, they are difficult to coagulate. Intrinsic colloids are another name for them. Starch, rubber, protein, and other materials are examples.
Lyophobic Colloids
A lyophobic colloid is one in which the dispersed phase has little or no affinity for the dispersion medium. The words lyo and phobic, respectively, denote "liquid" and "fear." As a result, they despise liquids. Because the dispersed phase does not readily form a colloid with the dispersion medium, they are difficult to manufacture and necessitate the employment of special techniques. They are brittle and require stabilising substances to stay alive. Extrinsic colloids are another name for them. Sols of metals such as silver and gold, as well as sols of metallic hydroxides, are examples.
Related Topics Link,
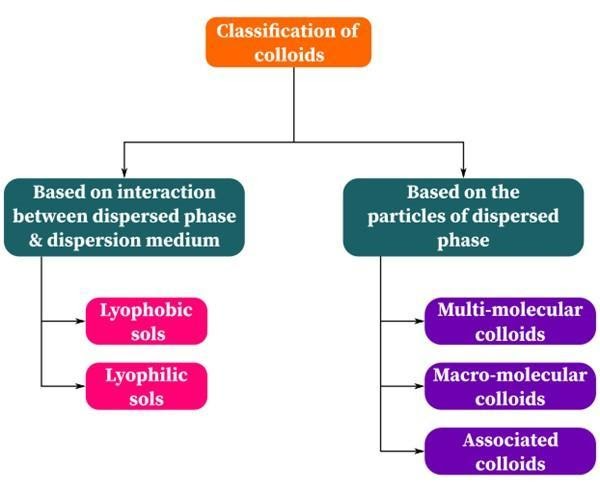
Types of Colloids Based on Type of Particles
The classification of colloids is done based on the types of colloids. The following are the categories:
- Multimolecular colloids
- Macromolecular colloids
- Associated colloids
Multimolecular Colloids
A solution created by the aggregation of a high number of atoms or tiny molecules (with diameters less than 1 nm) in a dispersed medium. Van der Waal forces hold the dispersed particles together.
Example:
Sulphur sol, and gold sol.
Macromolecular Colloids
Molecules with extremely high molecular weights combine to form macromolecules, which are exceedingly large molecules. The resulting colloidal solutions are known as macromolecular colloids when such compounds are dispersed in a suitable dispersion medium. As a result, macromolecular colloids have a large molecular mass. Lyophilic colloids are often macromolecular in nature.
Natural macromolecules, including starch, proteins, gelatin, cellulose, nucleic acids, and others, as well as synthetic polymers like polyethylene, polypropylene, and synthetic rubber, create macromolecular colloids when dispersed in suitable solvents.
Associated Colloids
When present in low quantities, some compounds behave as strong electrolytes, yet when present in high concentrations, they behave as colloidal sols. Particles agglomerate and demonstrate colloidal behaviour at greater concentrations. Micelles are microscopic clumps of collected particles. Associated colloids are another name for them. Micelles are formed above a certain temperature, known as the Kraft temperature (Tk), as well as a certain concentration, known as the critical micelle concentration. By diluting the colloids, they can be converted. Soaps and synthetic detergents are a couple of examples of related colloids.
Classification of Colloids Based on the Interaction Medium
Colloids can also be classed according on how the dispersed phase interacts with the medium:
Hydrophilic colloids: Colloids that love or are attracted to water are known as hydrophilic. Reversible sols are another name for them.
Agar, gelatin, and pectin are some examples.
Hydrophobic Colloid: These are the polar opposite of hydrophobic and repel water. Irreversible sols are another name for these.
Gold sols with clay particles, for example.
Colloids are characterised as lyophilic or lyophobic depending on the nature of the interaction between the dispersion medium and the dispersed phase.
Also read -
Some Solved Examples
Question.1 Which of the following is an example of a colloidal solution?
a) Salt solution
b) Air
c) Milk
d) Sand in water
Solution:
Milk is a colloidal solution where fat droplets are dispersed in water. The particles in milk are of colloidal size and do not settle out easily, making it a true colloid. Salt solution is a true solution, air is a mixture of gases (not colloidal), and sand in water is a suspension.
Hence, the correct answer is option (c)
Question.2 Which of the following is true for colloidal particles?
a) They are smaller than molecules.
b) They settle under the influence of gravity.
c) They are visible to the naked eye.
d) They cannot be separated by filtration.
Solution:
Colloidal particles are larger than molecules but smaller than particles in a suspension. They do not settle under gravity and cannot be separated by simple filtration, but they can be separated by methods like ultrafiltration.
Hence, the correct answer is option (d)
Question.3 What is the main characteristic that distinguishes a colloid from a solution?
a) Colloids are heterogeneous, while solutions are homogeneous.
b) Colloids have larger particles than solutions.
c) Colloids scatter light (Tyndall effect), while solutions do not.
d) All of the above
Solution:
Colloids are heterogeneous mixtures, while solutions are homogeneous. Colloids have larger particles that are small enough to remain suspended but large enough to scatter light, which is known as the Tyndall effect. Solutions have particles that are too small to scatter light effectively.
Hence, the correct answer is option (D)
Question 4: Which of the following is an example of an associated colloid?
(A) Starch solution
(B) Gold sol
(C) Soap solution
(D) Smoke
Solution:
Associated colloids form micelles at higher concentrations.
Soap molecules aggregate to form micelles above the critical micelle concentration (CMC).
- Starch solution → macromolecular colloid
- Gold sol → multimolecular colloid
- Smoke → aerosol
Hence, the correct answer is option (C)
Question 5: Which of the following colloidal systems has liquid as dispersed phase and gas as dispersion medium?
(A) Foam
(B) Aerosol
(C) Emulsion
(D) Gel
Solution:
When liquid droplets are dispersed in a gas, the colloid formed is called an aerosol.
Example:
- Fog
- Clouds
- Mist
Hence, the correct answer is option (B)
Practice More Questions With The Link Given Below
Frequently Asked Questions (FAQs)
1. Sols (Solid in liquid):
- Paint: Solid pigment particles dispersed in a liquid.
- Ink: Pigment or dye particles dispersed in water or other solvents.
- Blood: Red blood cells (solid) dispersed in plasma (liquid).
2.Gels (Liquid in solid):
- Gelatin: A liquid (water) dispersed in a solid matrix (gelatin).
- Agar-agar: A gel used in laboratories, made by dispersing water in a solid.
- Jelly: A colloidal gel with liquid dispersed in the solid matrix.
3. Emulsions (Liquid in liquid):
- Milk: Fat droplets dispersed in water.
- Mayonnaise: Oil droplets dispersed in water with the help of an emulsifier.
- Butter: Water droplets dispersed in fat.
4. Aerosols (Solid or liquid in gas):
- Fog: Water droplets dispersed in the air.
- Smoke: Solid particles dispersed in the air.
- Hairspray: Liquid droplets dispersed in air.
5. Foams (Gas in liquid or solid):
- Whipped cream: Air dispersed in cream (liquid foam).
- Soap foam: Air bubbles dispersed in a liquid.
- Foam rubber: Air bubbles dispersed in a solid matrix.
6. Solid Foams (Gas in solid):
- Pumice stone: Air pockets trapped within the solid matrix of volcanic rock.
- Aerogels: Solid materials with gas dispersed within them.
Multimolecular colloid particles are small collections of atoms and molecules with a dimension of less than one nanometre. They also have weak van der Waals interactions between particles, as opposed to macromolecular colloids, which have a significant molecular mass. Between macromolecular particles, they have strong chemical connections. Associated colloids differ from multimolecular and macromolecular colloids in that they form aggregated particles at high concentrations, giving them colloidal qualities. Because of their high molecular mass, they behave like macros.
These are the differences between multimolecular and macromolecular colloids.
Associated colloids are micro heterogeneous colloids in which a substance dissolved in the dispersion medium forms the micelles. They operate like a conventional strong electrolyte at low concentrations, but at greater concentrations, they exhibit colloidal features due to the production of aggregated particles.
With related colloids, two terms are used:
Specific Concentration: Micelles can also develop above a certain concentration, known as the critical micelle concentration.
A solution created by the aggregation of a high number of atoms or tiny molecules (with diameters less than 1 nm) in a dispersed medium. Van der Waals forces hold the dispersed particles together
Multimolecular Example:
Sulphur sol and gold sol.
A colloidal dispersion is made up of particles that are scattered in a continuous phase of solid, liquid, or gas (solid, liquid or gas). Colloidal particles are defined as particles having at least one dimension ranging from 1nm to 1m. Solid-liquid (suspensions), liquid-liquid (emulsions), gas-liquid (foams), and solid-gas (aerosols) dispersions are the most prevalent colloidal dispersions.
Because colloidal dispersion seeks to reduce surface energy, it is intrinsically thermodynamically unstable. As a result, a colloidal system's stability is inextricably related to a concept of time, which is defined by the process, usage, and/or application involved.

