Value of Electron - Charge & Mass of Electron
The electron is one of the most important fundamental particles in physics and plays a crucial role in understanding atomic structure, electricity, and modern electronics. Represented by the symbol e, an electron carries a negative electric charge and has an extremely small mass compared to protons and neutrons. Knowing the value of electron charge and electron mass is essential for studying concepts such as electric fields, electrostatics, current electricity, and quantum mechanics. The electron charge is a fundamental physical constant, while its mass is often expressed in kilograms as well as in electron volts (eV) using mass energy equivalence. In this article, we will explore the value of an electron, its charge, mass, electron volt, and its importance in physics.
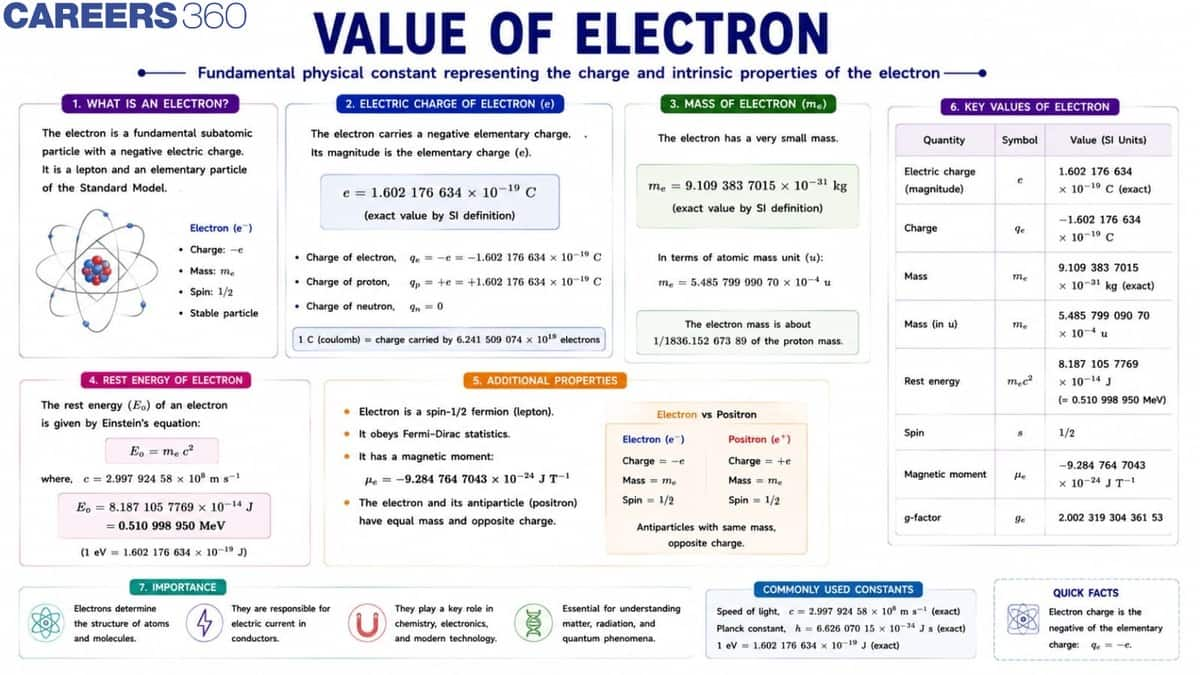
What is the Value of an Electron?
An electron is a fundamental subatomic particle represented by the symbol $\mathbf{e}$. It carries a negative electric charge and is one of the basic constituents of atoms. Important properties of an electron include its charge, mass, and spin, which are essential in understanding atomic structure, electricity, and quantum mechanics.
Value of Electron Charge
The elementary charge of an electron is:
$
e=1.60217662 \times 10^{-19} \mathrm{C}
$
The charge is negative in nature:
$
\text { Charge of electron }=-1.602 \times 10^{-19} \mathrm{C}
$
Value of Electron Mass
The mass of an electron is much smaller than that of a proton. It is approximately $\mathbf{1} / \mathbf{1 8 3 6}$ times the mass of a proton.
$
m_e=9.10938356 \times 10^{-31} \mathrm{~kg}
$
Electron Volt (eV)
An electron volt (eV) is the amount of kinetic energy gained by an electron when it is accelerated through a potential difference of one volt in vacuum.
$
1 \mathrm{eV}=1.6 \times 10^{-19} \mathrm{~J}
$
Since,
$
1 \mathrm{erg}=10^{-7} \mathrm{~J}
$
Therefore,
$1 \mathrm{eV}=1.6 \times 10^{-12} \mathrm{erg}$
|
Related Topics Link, |
Also Read:
- NCERT solutions for Class 12 Physics Chapter 1 Electric Charges and Fields
- NCERT Exemplar Class 12 Physics Solutions Chapter 1 Electric Charges and Fields
- NCERT notes Class 12 Physics Chapter 1 Electric Charges and Fields
Electron Mass in eV
In particle physics, the mass of an electron is often expressed in terms of its rest energy using Einstein's equation:
$E=m c^2$
The rest mass energy of an electron is:
$m_e c^2=0.511 \mathrm{MeV}$
This means the mass of an electron is equivalent to 0.511 mega electron volts (MeV) of energy.
Value of Charge of Electron
The electric charge of an electron is a fundamental physical constant. Electric charge is quantized, which means it exists in discrete units of the elementary charge $e$.
$e=1.6 \times 10^{-19} \mathrm{C}$
The charge of an electron is:
$e=-1.6 \times 10^{-19} \mathrm{C}$
The charge of a proton is:
$
+e=+1.6 \times 10^{-19} \mathrm{C}
$
Also check-
- NCERT Exemplar Class 11th Physics Solutions
- NCERT Exemplar Class 12th Physics Solutions
- NCERT Exemplar Solutions for All Subjects
NCERT Physics Notes:
Frequently Asked Questions (FAQs)
Atoms, which contain protons, neutrons, and electrons, make up all matter. Electrons are subatomic particles, they are incapable of being damaged, and they have three basic characteristics:
They revolve around an axis in the same way that planets do.
An electron has a rest mass of electron or weight of electron of 9.1 x 10 - 31 kg
Charge of 1 electron: Electrons are negatively charged in nature, with an electric charge of one electron of 1.6 x 10-19 C, the standard unit of charge of one electron for subatomic particles.
Because electrons have a rest the mass of an electron is of 9.1 x 10 - 31, . When an electron begins to move at the speed of light, the speed of the electron (v) equals the speed of light (c). The electron's the mass of an electron is would begin to vary, and these variations would be endless.
In kilograms, an electron has a mass of electron is of 9.1 x 10 - 31 kg. Because 1 kilogram equals 1000 g
As a result, an electron's mass of electron in grams is 9.1 x 10 -28 g.
An electron is a subatomic light particle (smaller than an atom) that carries a single unit of negative electricity, denoted by the letter e. Electrons can be either free (not linked to the atom) or bound to the nucleus. The magnitude of the elementary charge of electron is equal to the charge of electron of the electron (e) The value of basic unit of charge of electron is 1.6 x 10-19 C when the sign is negative (-e). As a result, electron charge value of electron value (- e) = -1.6 x 10-19 Coulomb.
The proton is a stable subatomic particle with a positive charge equal to that of an electron and a rest mass of proton is 1.67262 10-27 kg, or 1,836 times the mass of electron of an electron.

