Bohr's Model Of An Atom
To overcome the drawback of Rutherford's model of atom and to explain the line spectrum of hydrogen, Neils Bohr, a Danish physicist in 1913 proposed a new model of atom based upon Planck's quantum theory. This new model is called Bohr's model of atom. The key concept of Bohr's theory is the concept of quantization of electronic energy. By quantization we mean that a quantity cannot change gradually and continuously to have any arbitrary value but change only abruptly and discontinuously to have certain definite or discrete values. Bohr postulated that electrons revolve around the nucleus in certain fixed circular orbits in which they can continue revolving without gaining or losing energy. This means that the energy of the electron cannot change continuously but can have only definite values. Thus, we can say that energy of an electron is quantized.
This Story also Contains
- Bohr's Model and its Postulates
- Also study:
- Solved Examples Based On (Bohr's Postulate)-
- Conclusion
In this article, we will cover the concept of Bohr's model and its postulates. This concept falls under the broader category of Atomic structure, which is a crucial chapter in Class 11 chemistry. It is not only essential for board exams but also for competitive exams like the Joint Entrance Examination (JEE Main), National Eligibility Entrance Test (NEET), and other entrance exams such as SRMJEE, BITSAT, WBJEE, BCECE, and more.
Also Read -
Bohr's Model and its Postulates
Neil Bohr was first to explain quantitatively the general features of hydrogen atom i.e., structure and its spectrum. He extended the work of Rutherford to give the structure of atom. Once, the existence of the nucleus was proved through Rutherford's model of atom, now the work left was to concentrate on the arrangement of electrons around the nucleus. Neil Bohr was successful in answering the questions like:
- Why line spectrum of atoms is obtained?
- The explanation of spectral lines of hydrogen atom etc.
Bohr’s model also gave some explanation for the phenomenon that is known as stationary states, whereby the electrons in their stable orbits do not radiate as perceived by classical theories. This model was able to thoroughly account for the spectrum of hydrogen and also paved the way for something that turned out to be quantum mechanics. Even though there are significant weaknesses in Bohr’s model like the inability to explain the phenomena of multielectron atoms, this model is considered one of the important steps in the development of atomic theory as it gives a proper understanding of the electron behavior in an atom.
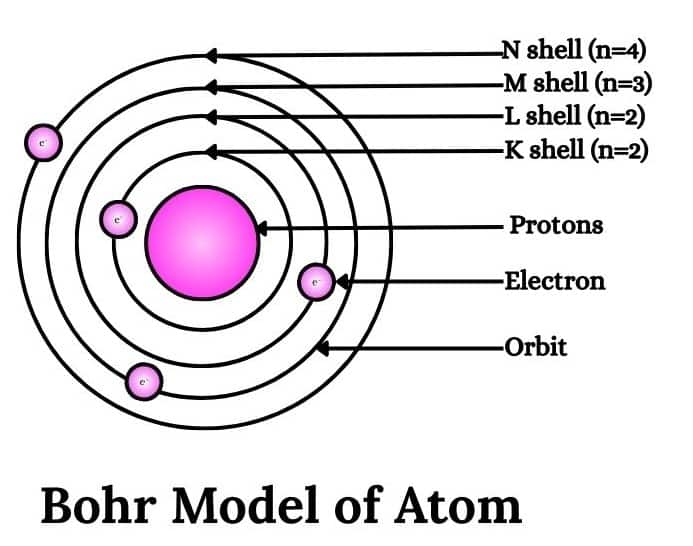
1. The electron in the hydrogen atom can move around the nucleus in a circular path of fixed radius and energy. These paths are called orbits, stationary states, or allowed energy states and are arranged concentrically around the nucleus. The force of attraction between the nucleus and an electron provides the centripetal force required by the electron to carry out the circular motion.
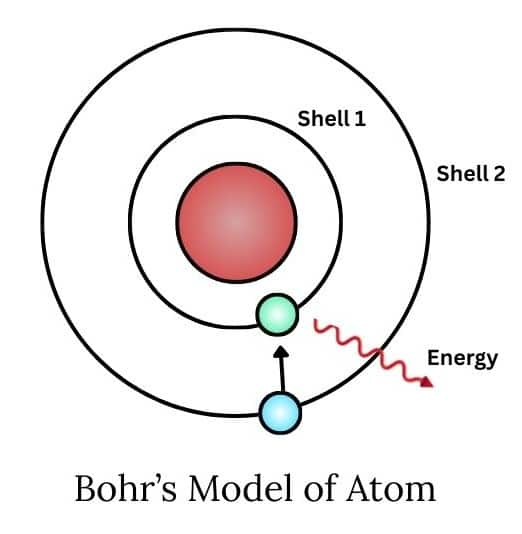
2. The energy of an electron in the orbit does not change with time. However, the electron will move from a lower stationary state to a higher stationary state when the required amount of energy is absorbed by the electron or energy is emitted when an electron moves from a higher stationary state to a lower stationary state.
The energy of the different stationary states in case of hydrogen atom are given by the expression (called Bohr formula):
3. Energy can be absorbed or emitted when an electron transitions between two different orbits, and the frequency of the photon involved can be calculated using the formula:
4. The angular momentum of an electron is quantized. In a given stationary state, it can be expressed as
So only those energy states (or orbits) are allowed in which the above equation holds for the angular momentum.
Important points:
- The radii of the stationary states of the hydrogen atom are given by the expression:
- The velocities of the electrons in different orbits are given by the expression:
Note: Bohr's model is only valid for Hydrogen species or electronic species that contain only a single electron.
Also study:
Recommended topic video on (Bohr's Model Of An Atom )
Solved Examples Based On (Bohr's Postulate)-
Example 1: Which of the following statements is incorrect for Bohr's model of an atom?
1) It is valid for a multi-electronic species
2) The angular momentum of an electron is quantized
3) The centripetal force of attraction required for circular motion is provided by the electrostatic force of attraction between the electron and the nucleus
4) Orbits have fixed energy and are referred to as stationary states
Solution:
Bohr's model is valid for only uni-electronic species.
All other given statements given in the options are correct.
Hence, the answer is the option (1).
Example 2:
Which of the following is incorrect for Bohr's model of an atom?
1) It is valid for multi-electronic species.
2) The angular momentum of an electron is quantized.
3) The centripetal force of attraction required for circular motion is provided by the electrostatic force of attraction between the electron and the nucleus.
4) Orbits have fixed energy and are referred to as stationary energy.
Solution:
Bohr's model is valid for electronic species only.
Hence, the answer is the option (1).
Example 3:
Assume that the radius of the first Bohr orbit of hydrogen atom is $0.6 Ao $ . The radius of the third Bohr orbit of
Solution:
Practice More Questions From the Link Given Below:
Conclusion
The version that was presented by Bohr was able to explain the spectral traces of hydrogen. Bohr’s model of the atom can be viewed as a scientifically critical stage in the paradigm shift from the old paradigm of classical physics to the new approaches of the modern quantum mechanics paradigm. While Bohr’s model had some problems, such as its inability to deliver a qualitative description of multielectron atoms and the quantization of angular momentum, it was useful in developing an early atomic model that helped accurately describe atomic spectra.
Also Check-
Frequently Asked Questions (FAQs)
According to Bohr, the electrons reside in a specified region in a circle which is stable and hence can be referred to as stationary circular orbits because they do not emit energy due to centripetal force balancing the attractive force of the nucleus.
From emission and absorption spectra, discrete electron transition.
As Bohr's model applies to Hydrogen atoms only, that's why this model couldn't explain the multielectron atoms.
The electron in the hydrogen atom can move around the nucleus in a circular path of fixed radius and energy. These paths are called orbits, stationary states, or allowed energy states and are arranged concentrically around the nucleus. The force of attraction between the nucleus and an electron provides the centripetal force required by the electron to carry out the circular motion.

