Important Chemical Reactions For CBSE Class 12 - Reaction and FAQs
The reactions in which some bonds get broken towards reactant molecules and form new bonds towards product molecules, which further form new substances. Reactants are those substances that are generally present on the left side, and products are present on the right-hand side. There are many types of chemical reactions out of which some important chemical reactions that you all studied in class 12 are defined as follows:
This Story also Contains
- Sandmeyer Reaction:
- Gattermann Reaction:
- Balz-Schiemann Reaction:
- Finkelstein Reaction:
- Swart's Reaction:
- Wurtz Reaction:
- Wurtz-Fitting Reaction:
- Fitting Reaction:
- Friedel-Crafts Alkylation Reaction:
- Frieldel-Crafts Acylation Reaction:
- Diazotisation Reaction
- Reimer-Tiemann Reaction:
- Kolbe’s reaction:
- Rosenmund Reaction:
- Clemmensen Reduction
- Wolf-Kishner Reaction
- Gattermann-Koch Reaction
- Sabatier-Sanderson Reaction
- Houben-Hoesch Reaction
- Carbylamine Reaction:
- Aldol Condensation
- Williamson Ether Synthesis
- Cannizzaro Reaction
- Gabriel Phthalimide Reaction

Sandmeyer Reaction:
This is the reaction that is generally used for the preparation of aryl halides from aryl diazonium salt, where aryl corresponds to the ring-like structure. The Sandmeyer reaction can also be called a substitution reaction of any aromatic ring by introducing diazonium salt, which is done through a displacement reaction, and copper is used as a catalyst in this reaction. Reaction can be shown as follows:
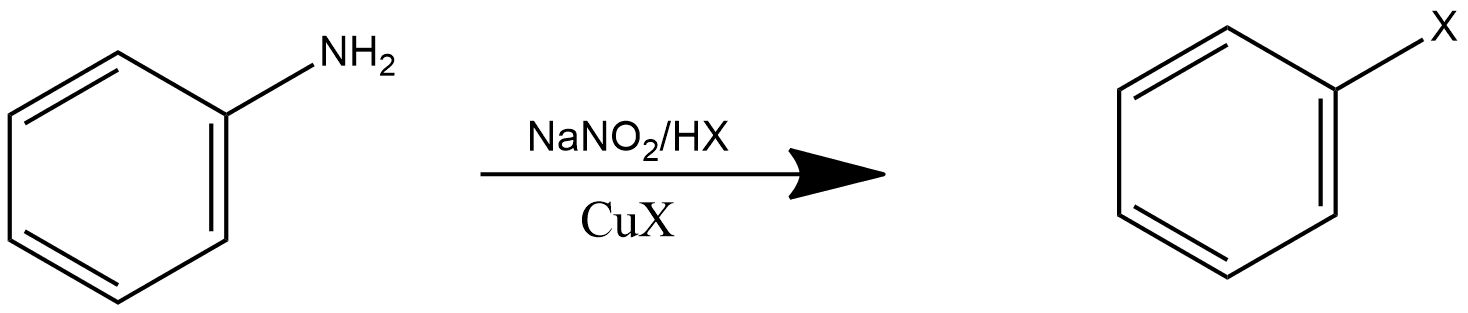
Here X represents a halogen, which can be chlorine, bromine, iodine, etc, or maybe a cyanide ion.
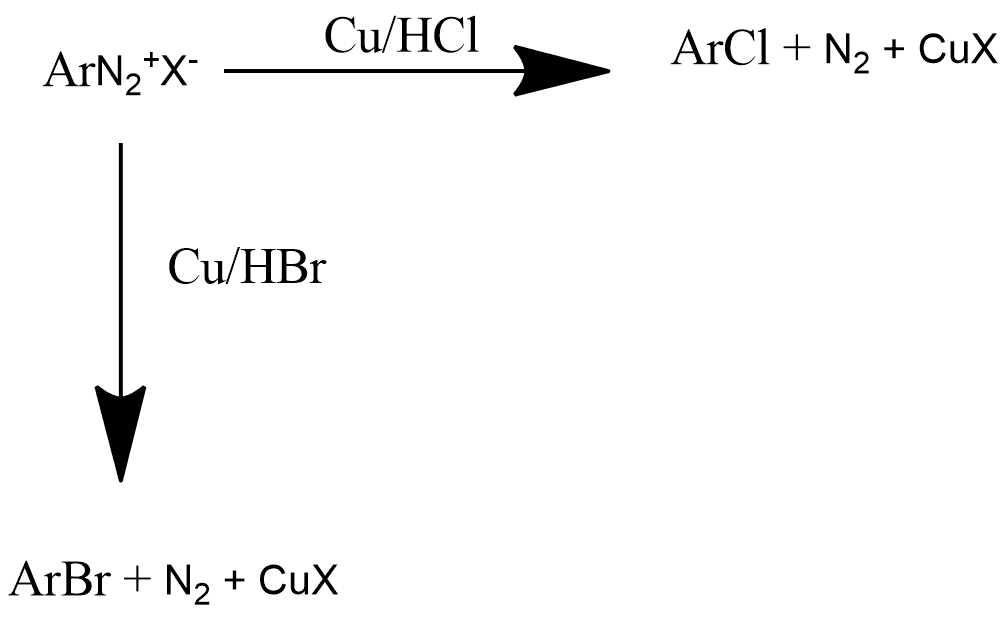
Gattermann Reaction:
Gattermann reaction is also known by the method of formylation of those compounds that have aromatic rings. It is very similar to Friedel-crafts reaction. This reaction is generally used for obtaining chloro or bromo benzene from bromobenzene diazonium chloride by reacting with Cu/HCl or Cu/HBr, respectively and the reaction can be represented as:
Balz-Schiemann Reaction:
This reaction is used for preparing aryl fluoride this can be done by a number of steps first arene-diazonium chloride is prepared with the help of fluoroboric acid then arene diazonium fluoroborate which is precipitated and decomposes further which produces aryl halide with the help of heating. Reaction can be shown as:

Finkelstein Reaction:
This reaction is mainly prepared for preparing alkyl iodides which can be proceeded by the reaction between alkyl chlorides with sodium iodide represented by the chemical formula NaI in the presence of dry acetone and reaction can be shown below:
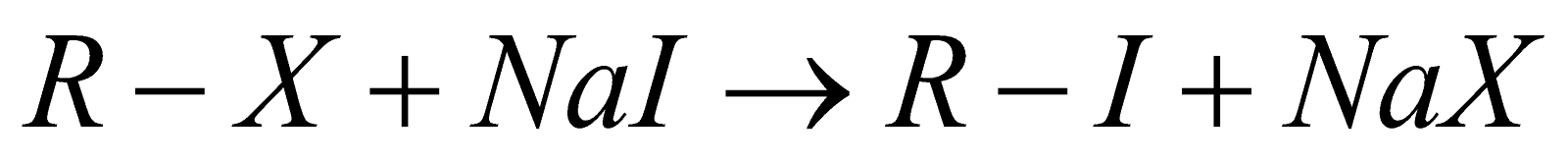
Here X represents halogen which can be chlorine, bromine, iodine, etc.
Swart's Reaction:
This is the reaction that gives alkyl fluoride as a product when alkyl chloride is heated in the presence of metallic fluoride compounds which can be represented as 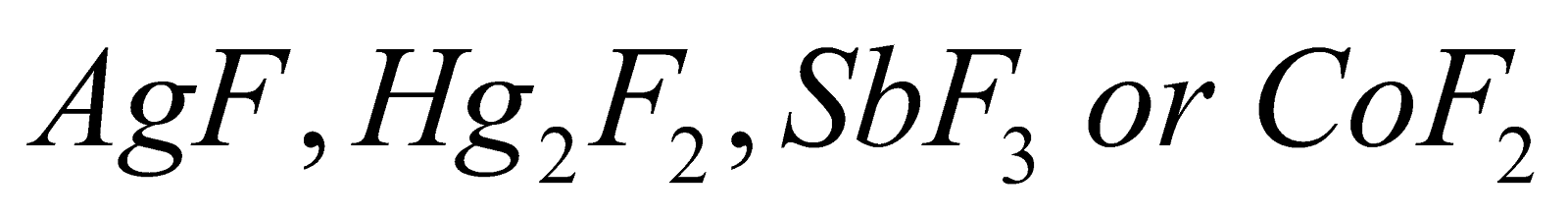 and this further gives alkyl fluorides. Reaction can be shown as:
and this further gives alkyl fluorides. Reaction can be shown as:
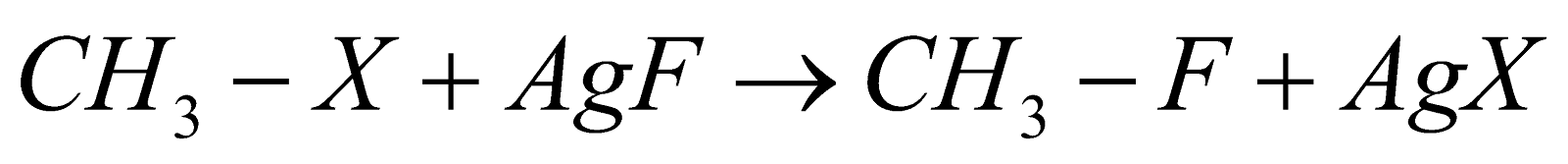
Here X represents a halogen, which can be chlorine, bromine, iodine, etc.
Wurtz Reaction:
This reaction is generally used for the production of hydrocarbons, in which alkyl halides generally react with sodium and dry ether in this we get double the number of carbon atoms that we have on the reactant side, and the reaction can be shown as follows:
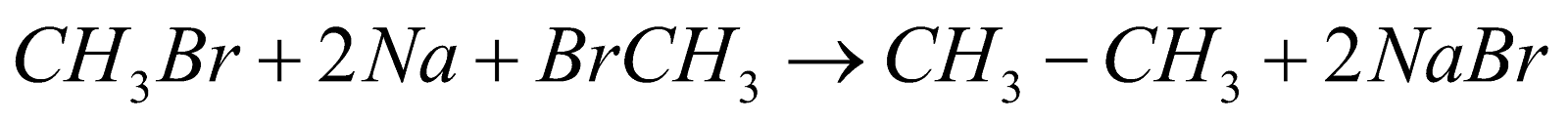
This reaction proceeds in the presence of dry acetone.
Wurtz-Fitting Reaction:
This reaction is one of the most basic and important reactions for getting alkyl arenes. In this reaction, alkyl halide or aryl halide reacts with sodium in the presence of dry ether, and the reaction can be represented as:
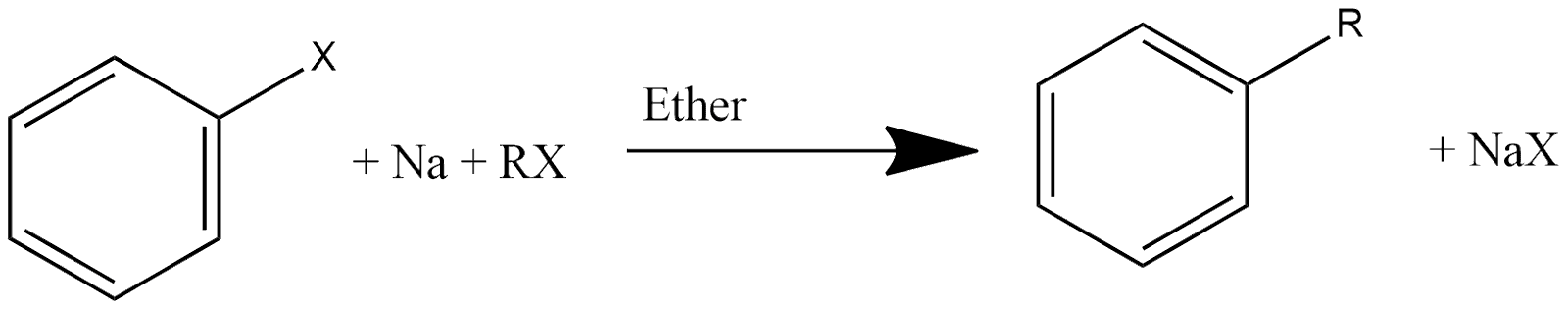
Fitting Reaction:
This is one of the reaction in which two aryl groups get joined with each other and we can define the reaction in such a way that aryl halides when react with sodium in dry ether which give analogous compounds in which two aryl groups are joined with each other. Reaction can be shown as:
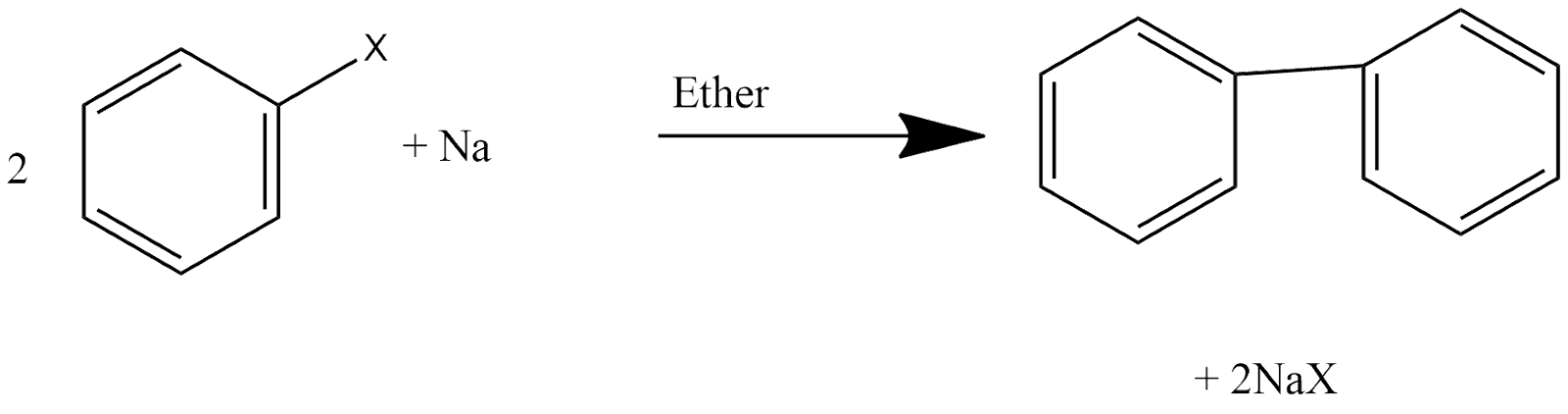
Friedel-Crafts Alkylation Reaction:
In this reaction benzene is prepared with the help of an alkyl halide in the presence of anhydrous aluminum chloride represented by the chemical formula AlCl3 which further gives the alkyl benzene as a product and the reaction can be shown as:

Frieldel-Crafts Acylation Reaction:
Friedel-Craft acylation is the same as Friedel alkylation. The difference is that in an acyl halide reacts with benzene, but an alkyl halide reacts with benzene, and both reactions take place in the presence of anhydrous aluminum chloride, and the reaction can be shown as:

Diazotisation Reaction
The diazotization reaction is a chemical reaction in which an aromatic amine (such as aniline) is converted into a diazonium salt using nitrous acid (HNO₂) or sodium nitrite (NaNO₂) in the presence of an acid (like hydrochloric acid, HCl) at low temperatures (0-5°C) to produce Benzene Diazonium Chloride.
.png)
Reimer-Tiemann Reaction:
This is a multistep reaction in which salicylaldehyde is the product of this reaction, which can be shown and explained as follows:
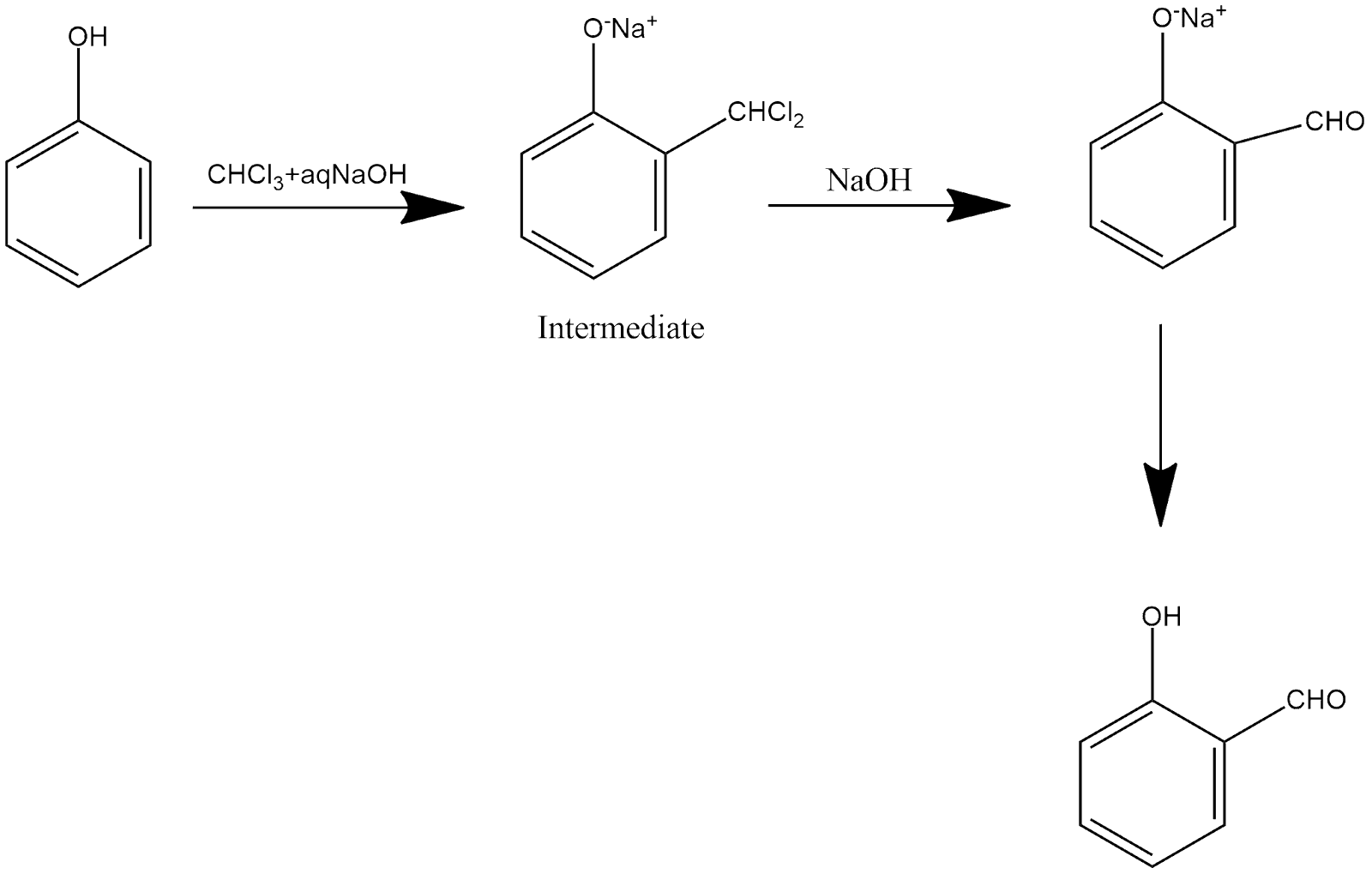
In a step, phenol is reacted with chloroform and sodium hydroxide and forming an intermediate compound, and further this reacts with sodium hydroxide, in which an aldehyde group is present at the ortho position, which on heating gives salicyladehyde.
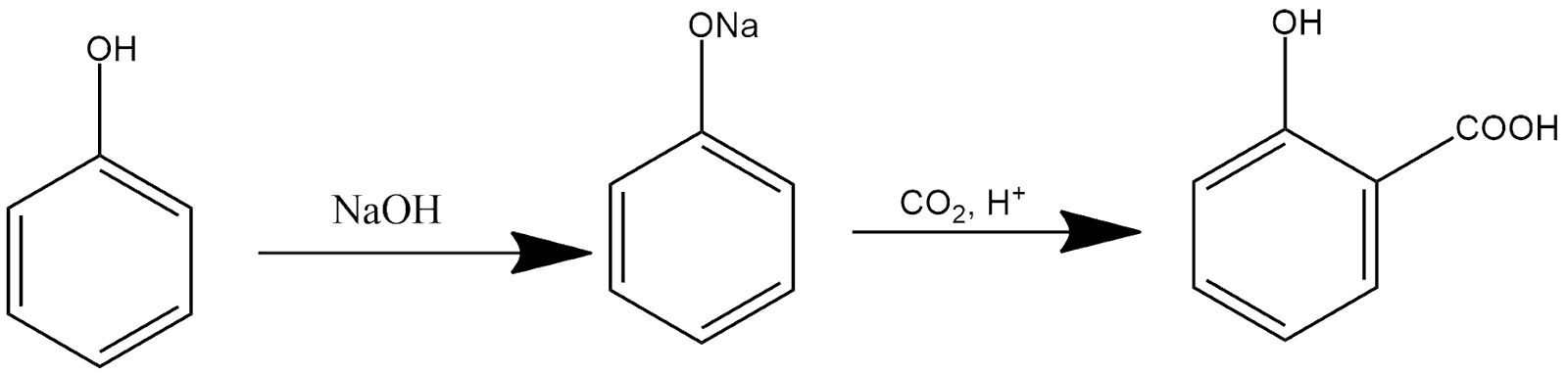
Kolbe’s reaction:
In the presence of sodium hydroxide phenol gives sodium phenoxide, which further reacts with carbon dioxide in an acidic medium and produces hydroxybenzoic acid as a product, and the reaction can be shown as:
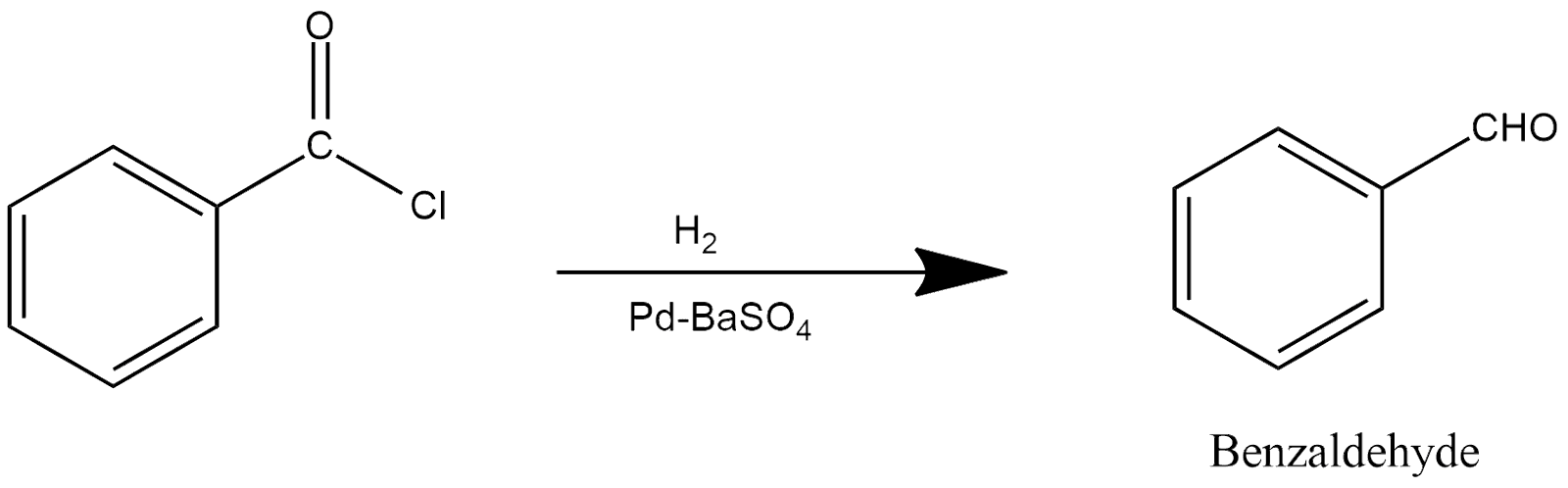
Rosenmund Reaction:
This reaction is generally performed in the presence of a catalyst, which is known as the name of Rosenmund catalyst, which be either in the form of palladium or barium sulfate, represented by PdSO4 or BaSO4. In this reaction, benzoyl chloride undergoes through hydrogenation process and produces benzaldehyde in the presence of Rosenmund catalyst, and the reaction can be shown as:
Clemmensen Reduction
The Clemmensen Reduction is a chemical reaction that involves the reduction of carbonyl compounds (such as aldehydes and ketones) to alkanes using zinc amalgam (Zn/Hg) in the presence of hydrochloric acid (HCl). It is an important method for reducing aromatic aldehydes and ketones to the corresponding alkanes and is especially used in organic synthesis.
.png)
Wolf-Kishner Reaction
The Wolf–Kishner reduction is a chemical reaction that involves the reduction of carbonyl compounds (such as aldehydes and ketones) to alkanes. This reduction is achieved using hydrazine (N₂H₄) in the presence of potassium hydroxide (KOH) and heat. The reaction is a powerful method for reducing aromatic and aliphatic aldehydes or ketones without affecting other functional groups
.png)
Gattermann-Koch Reaction
This reaction also produces benzaldehyde, in which benzene reacts with carbon monoxide and hydrochloric acid in the presence of aluminium chloride, and gives benzaldehyde as a product, which can be shown as:
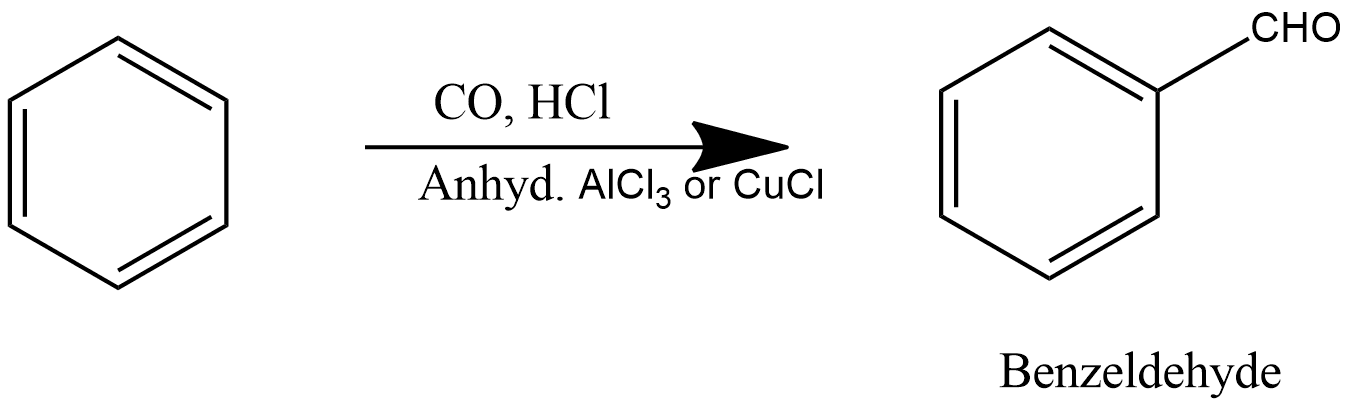
Sabatier-Sanderson Reaction
In this reaction, the hydrogenation process occurs, i.e. addition of hydrogen atoms, and this reaction converts an alkene into an alkane. This is one of the main reactions by which ethane is prepared on at large scale in Ethylene or ethane undergoes the process of hydrogenation in the presence of the catalyst Ni and gives ethane. The reaction can be represented as:
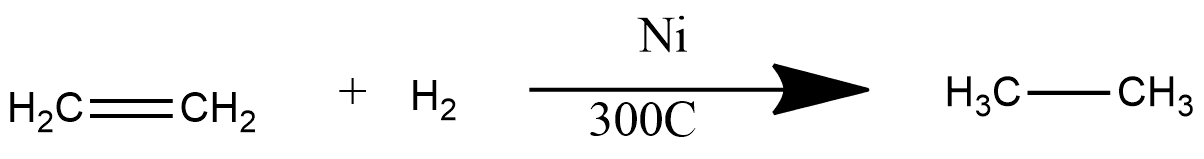
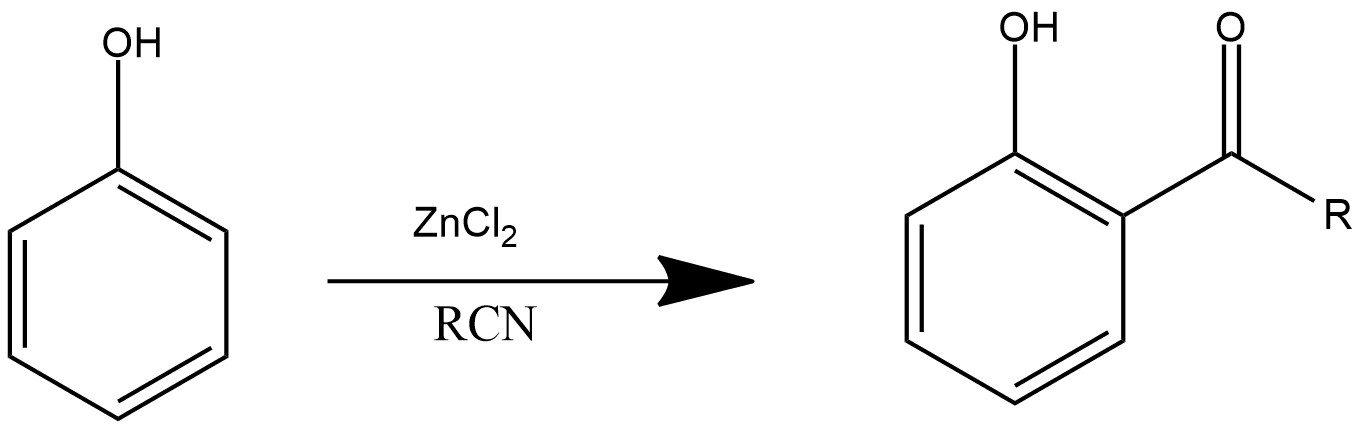
Houben-Hoesch Reaction
This is one of the important reaction that provide acyl chlorides as its product. This is very similar to Friedel-Craft alkylation and Friedel-Craft acylation, as we discussed above in this reaction, phenol reacts with zinc chloride and cyanide group, which further produces more than one acyl group in the product, and the reaction can be shown as:
Carbylamine Reaction:
This is the reaction which is used to produce isocyanides or carbylamines as a product, which is why they are named as carbylamines reaction. Aliphatic and aromatic amines are heated in the presence of chloroform and ethanolic potassium hydroxide, and produce isocyanides during this reaction
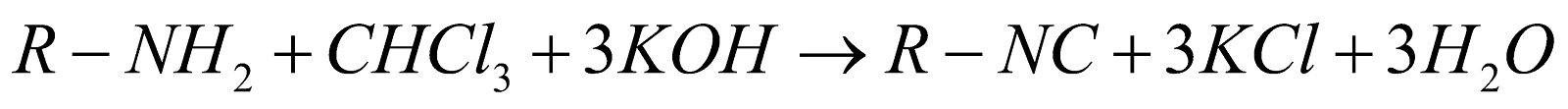
Other than these reactions, many other reactions are also known to u,s which are aldol condensation, cross aldol condensation, Gattermann-Koch synthesis, Hoffmann bromamide reaction, HVZ reaction, Fehling test, Tollen’s reagent test, etc.
Aldol Condensation
Aldol condensation is an organic reaction in which two molecules of aldehydes or ketones (having at least one $\alpha$-hydrogen) react in the presence of a dilute base (like NaOH ) to form a $\beta$-hydroxy aldehyde (aldol) or $\beta$-hydroxy ketone. On heating, this product loses water (dehydration) to form an $\alpha, \beta$-unsaturated aldehyde or ketone.
Aldol Addition
$2 \mathrm{CH}_3 \mathrm{CHO} \xrightarrow{\mathrm{NaOH}} \mathrm{CH}_3 \mathrm{CH}(\mathrm{OH}) \mathrm{CH}_2 \mathrm{CHO}$
Aldol condensation
$\mathrm{CH}_3 \mathrm{CH}(\mathrm{OH}) \mathrm{CH}_2 \mathrm{CHO} \xrightarrow{\Delta} \mathrm{CH}_3 \mathrm{CH}=\mathrm{CHCHO}+\mathrm{H}_2 \mathrm{O}$
Williamson Ether Synthesis
The Williamson Ether Synthesis is a laboratory method used to prepare ethers by reacting an alkoxide ion with a primary alkyl halide.
$\mathrm{R}-\mathrm{O}^{-} \mathrm{Na}^{+}+\mathrm{R}^{\prime}-\mathrm{X} \rightarrow \mathrm{R}-\mathrm{O}-\mathrm{R}^{\prime}+\mathrm{NaX}$
$\mathrm{CH}_3 \mathrm{ONa}+\mathrm{C}_2 \mathrm{H}_5 \mathrm{Br} \rightarrow \mathrm{CH}_3 \mathrm{O}-\mathrm{C}_2 \mathrm{H}_5+\mathrm{NaBr}$
Cannizzaro Reaction
The Cannizzaro Reaction is a redox reaction in which non-enolizable aldehydes (aldehydes without α-hydrogen) undergo self-oxidation and reduction when treated with a strong base (like concentrated NaOH or KOH).
$2 \mathrm{RCHO} \xrightarrow{\text { Conc. } \mathrm{NaOH}} \mathrm{RCOONa}+\mathrm{RCH}_2 \mathrm{OH}$
Where:
One aldehyde molecule → Oxidised → Carboxylate ion
Second aldehyde → Reduced → Alcohol
Gabriel Phthalimide Reaction
It is a method used for the preparation of primary aliphatic amines (1° amines) from phthalimide.
Reagents Needed
- Phthalimide
- KOH (forms potassium phthalimide)
- Alkyl halide (R–X)
- Hydrazine or NaOH (for final release)
Step 1 - Formation of Potassium Phthalimide
$\text { Phthalimide }+\mathrm{KOH} \rightarrow \text { Potassium Phthalimide }+\mathrm{H}_2 \mathrm{O}$
Step 2 - Nucleophilic Substitution (SN2 Reaction)
$\text { Potassium Phthalimide }+\mathrm{R}-\mathrm{X} \rightarrow \mathrm{~N} \text {-Alkyl Phthalimide }+\mathrm{KX}$
Step 3 - Hydrolysis / Hydrazinolysis
$\mathrm{N} \text {-Alkyl Phthalimide } \xrightarrow{\mathrm{NaOH} / \mathrm{H}_2 \mathrm{O}} \mathrm{RNH}_2+\text { Phthalic Acid Salt }$
Also Read
| Pseudo First Order Reaction |
| First Order Reactions |
| Zero Order Reaction |
| Rate of Reaction |
| Second Order Reaction |
Also Read:
Frequently Asked Questions (FAQs)
A chemical reaction is a process that involves the rearrangement of atoms and molecules to form new substances. It involves the breaking and forming of chemical bonds.
A chemical equation is a symbolic representation of a chemical reaction using chemical formulas and symbols. It shows the reactants, products, and their relative amounts. For example: 2H₂ + O₂ → 2H₂O
Hell Volhard Zelinsky reaction.
Tollen’s test is basically known by the name silver mirror test in which when aldehyde gets treated with freshly prepared ammoniacal silver nitrate then it produce a mirror i.e. a bright silver mirror which represents the formation of a silver mirror.
When an aldehyde is heated with fehling solution then it produces reddish brown color precipitates.
Aldol reaction is done by those aldehydes and ketones which have one alpha hydrogen and it goes through the reaction in the presence of dilute alkali which acts as a catalyst and produce beta hydroxy aldehydes and beta hydroxyl ketones.

