This chapter is asked in board and competitive examinations with focus on preparation methods, chemical properties, and reaction mechanisms of these organic compounds.
Alcohols, Phenols and Ethers
Do you know why perfumes have aroma, what gives hand sanitisers their disinfecting powers, why ethanol is used as fuel in vehicles and what makes alcohol act as antiseptic? The answer to all such questions lies in Alcohols, phenols and Ethers. A special class of compounds that feature oxygen-containing organic compounds. When the OH group replaces the hydrogen atom in aliphatic hydrocarbons alcohols are formed the same goes for phenols when the OH group replaces the hydrogen of aromatic hydrocarbons then Phenols are formed while Ethers are formed when the alkoxy (R-O) or aryloxy (Ar-O) group substitutes the hydrogen atom in hydrocarbons. Refer to this article of Chemistry to understand these concepts.
This Story also Contains
- Important Topics
- Overview Of The Chapter
- Study Links
- Alcohols, Phenols and Ethers in Different Exams
- Important PYQs
- Importance Of Alcohols, Phenols, And Ethers In Class 12:
Important Topics
Given below the topics and subtopics of the chapter that are most important for examinations and quick revision.
Preparation Of Alcohols:
Alcohols are compounds that have attached one or more (-OH) hydroxyl groups to them. The alcohols do not occur in the free form in nature. They are obtained from flowers, leaves, and stems of plants. The Preparation Of Alcohols includes many methods such as the hydrolysis of halides, hydration of alkenes, Grignard reagents, etc
Physical And Chemical Properties Of Alcohols:
Physically, alcohols have higher boiling and melting points than alkanes and alkenes of similar molecular weight due to the presence of a strong hydrogen bond among them. Chemically, alcohols undergo acylation, whereby an acyl group is introduced into the molecule, and oxidation, whereby alcohol is oxidized into aldehydes, ketones, or carboxylic acids. There are many other Physical And Chemical Properties Of Alcohol.
Preparation Of Phenols:
The preparation of phenol in most cases involves the hydroxylation of an aromatic compound, usually benzene. Phenol is manufactured from the hydrocarbon, cumene. Cumene (isopropylbenzene) is oxidized in the presence of air to cumene hydroperoxide.
Physical And Chemical Properties Of Phenols:
Phenols are colorless liquids or white crystalline solids. The boiling points of phenols are much higher than the corresponding aromatic hydrocarbons and the haloarenes. Chemically, Phenols contain both a hydroxyl group and an aromatic ring; therefore, they are highly prone to various chemical reactions. In the Reimer-Tiemann reaction, phenols form ortho-hydroxybenzaldehyde upon formylation with chloroform and base. There are other many Physical And Chemical Properties Of Phenols.
Williamson's Ether Synthesis:
Williamson's Ether Synthesis is an old method in organic chemistry for the synthesis of ethers. The reaction involves the nucleophilic substitution by an alkoxide ion, RO-, by a primary alkyl halide $\mathrm{R}^{\prime}-\mathrm{X}$, to form an ether, R-O-R.
Overview Of The Chapter
This chapter provides a concise understanding of the main concepts, reactions, and applications covered, helping students build a strong foundation:
Classification Of Alcohols And Phenols
-
Alcohols and phenols are classed as monohydric, dihydric, trihydric, or polyhydric depending on how many hydroxyl groups they have in their molecules: one, two, three, or many, respectively
-
Alcohols with the OH group connected to a primary, secondary, or tertiary carbon atom are known as primary, secondary, and tertiary alcohols.
-
Allylic alcohol is made up of an -OH group attached to an $\mathrm{sp}^3$ Hybridised carbon next to a carbon-carbon double bond, whereas benzylic alcohol is made up of an -OH group attached to an $\mathrm{sp}^3$ hybridized carbon close to an aromatic ring. Alcohols having -OH group bonded to carbon-carbon double bond is called vinylic alcohol
Structure Of Alcohols And Phenols
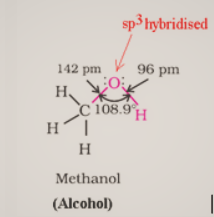
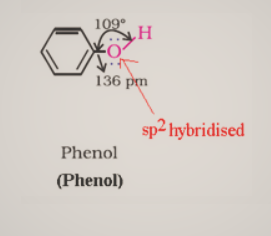
Alcohols have $\mathrm{sp}^3$ hybridized oxygen atoms and hybrid atomic orbitals in a tetrahedral configuration. Due to lone pair repulsion, this angle for of methyl alcohol is (C – O – H) 108.9°.The -OH group in phenols is connected to $\mathrm{sp}^2$ hybridized carbon, giving the C – O bond a partial double bond nature.
Preparation Of Alcohols
-
From alkenes
This reaction is in accordance with markovnikov's rule, through acid-catalyzed hydration.
$\mathrm{CH}_3 \mathrm{CH}=\mathrm{CH}_2+\mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{CH}_3-\mathrm{CH}(\mathrm{OH})-\mathrm{CH}_3$
-
By hydroboration-oxidation
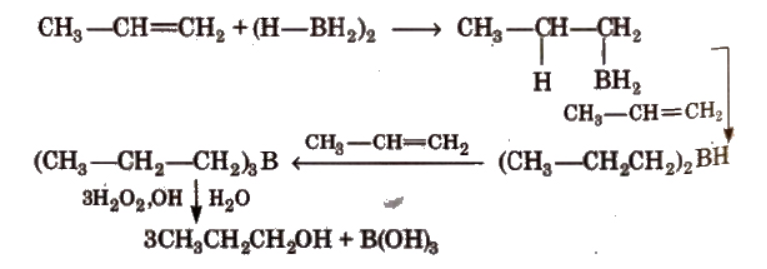
-
By reduction of aldehydes and ketones
$\mathrm{RCHO}+\mathrm{H}_2 \rightarrow \mathrm{R} \mathrm{CH}_2 \mathrm{OH}$
$\mathrm{RCOR} \xrightarrow{\mathrm{NaBH}_4} \mathrm{R}-\mathrm{CH}(\mathrm{OH})-\mathrm{R}$
-
By reduction of carboxylic acids
$\mathrm{RCOOH}^2 \xrightarrow{\mathrm{LiAlH}_4 \mathrm{H}_2 \mathrm{O}} \mathrm{ROH}$
- From Grignard reagents
$\begin{aligned} & \mathrm{HCHO}+\mathrm{RMgX} \xrightarrow{\mathrm{H}_2 \mathrm{O}} \mathrm{RCH}_2 \mathrm{OH}+\mathrm{Mg}(\mathrm{OH}) \mathrm{X} \\ & \mathrm{RCHO}+\mathrm{RMgX} \xrightarrow{\mathrm{H}_2 \mathrm{O}} \mathrm{R}-\mathrm{CH}(\mathrm{R}) \mathrm{OH}+\mathrm{Mg}(\mathrm{OH}) \mathrm{X} \\ & \mathrm{RCOR}+\mathrm{RMgX} \xrightarrow{\mathrm{H}_2 \mathrm{O}} \mathrm{R}-\mathrm{C}(\mathrm{R})(\mathrm{R}) \mathrm{OH}+\mathrm{Mg}(\mathrm{OH}) \mathrm{X}\end{aligned}$
Physical Properties Of Alcohols And Phenols
Lower alcohols are colorless liquids, $\mathrm{C}_5-\mathrm{C}_{11}$ alcohols are oily liquids, and $\mathrm{C}_{12}$ and higher alcohols are waxy solids. Alcohols are miscible with water because their hydroxyl groups can form H-bonds with water. With increasing molecular mass, solubility decreases. Because polar molecules have intermolecular hydrogen bonding, the boiling points of alkanes are greater than expected.
These are colorless liquids or crystalline solids that turn coloured over time due to gradual oxidation in the presence of air. Carboxylic acids are another name for phenol. Phenols establish intermolecular H-bonds with other phenol molecules and with water due to the presence of a polar -OH bond.
Ether:
Ether is a pleasant-smelling colourless volatile liquid that is highly flammable. It is used as an anesthetic and as a solvent or intermediate in industrial processes.
Preparation Of Ether
-
Dehydration of alcohols
.png)
-
Williamson synthesis
$\mathrm{R}-\mathrm{X}+\mathrm{R}-\mathrm{O}-\mathrm{Na} \rightarrow \mathrm{R}-\mathrm{O}-\mathrm{R}+\mathrm{NaX}$
Physical Properties Of Ethers
Since ethers' C-O bonds are polar, they have a net dipole moment. Their boiling points are equivalent to alkanes with similar molecular weights, although they are lower than alcohols. It's because ethers don't have H-bonding. Ether miscibility with water is similar to that of alcohols of the same molar mass. It's because others, like alcohol, can make H-bonds with water.
Study Links
Given below some links to access the important resources for better understanding of the chapter:
Alcohols, Phenols and Ethers in Different Exams
| Exam Name | Focus Area | Common Topics Asked | Preperation Tips |
| CBSE Board | Conceptual understanding | Preparation, properties, reactions | Revise NCERT reactions and examples |
| JEE Main | Concept application | Acidity of phenols, reactions of alcohols | Practise MCQs and conversions |
| JEE Advanced | Analytical & mechanism-based | Reaction mechanisms, rearrangements | Focus on detailed mechanisms |
| NEET | Basic concepts | Uses, reactions, IUPAC naming | Study NCERT line by line |
| State Board Exams | Theory-oriented | Definitions, preparation methods | Learn key reactions and properties |
| Chemistry Olympiads | Advanced application | Reaction mechanisms, comparative reactivity | Practise advanced organic problems |
Important PYQs
This section includes frequently asked questions from previous years that help in understanding exam patterns:
Question: 1 Given below are two statements:
Statement (I): The boiling points of alcohols and phenols increase with increase in the number of C -atoms.
Statement (II): The boiling points of alcohols and phenols are higher in comparison to other class of compounds such as ethers, haloalkanes.
In the light of the above statements, choose the correct answer from the options given below:
(1) Both Statement I and Statement II are false
(2) Statement I is false but Statement II is true
(3) Statement I is true but Statement II is false
(4) Both Statement I and Statement II are true
Answer:
B.P. $\propto$ M.W.
Therefore, the boiling points of alcohols and phenols increase with the increase in the number of C-atoms.
B.P. $\propto$ Intermolecular hydrogen bonding
Alcohol \& Phenol have intermolecular H-bonding
Therefore, the boiling points of alcohols and phenols are higher in comparison to other classes of compounds, such as ethers and haloalkanes.
Hence, the correct answer is option (4).
Question: 2 Grignard reagent reacts with aldehyde to form alcohol. How many moles of $\mathrm{CH}_3 \mathrm{MgBr}$ are required to react with one mole of formaldehyde and the type of alcohol produce in respectively
(1) 1, Primary alcohol
(2) 1, Secondary alcohol
(3) 2, Secondary alcohol
(4) 2, Tertiary alcohol
Answer:
$\mathrm{CH}_3 \mathrm{MgBr}+\mathrm{HCHO}^\xrightarrow{\mathrm{H}_2 \mathrm{O}} \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{OH}+\mathrm{HOMgBr}$
So, 1 mole of $\mathrm{CH}_3 \mathrm{MgBr}$ is required and a primary alcohol is formed.
Hence, the answer is the option (1).
Practice more questions from the link given below
For more questions to practice ,following MCQs will help in preparation of competitive examinations:
Importance Of Alcohols, Phenols, And Ethers In Class 12:
Alcohol and ether are classes of organic compounds that find wide usage in industries as well as for domestic purposes. Alcohol is formed when a saturated carbon atom is bonded to a hydroxyl (-OH) group. Ether is formed when an oxygen atom is connected to two alkyl or aryl groups. The main concepts include the classification of alcohol and ether, preparation and important reactions. Alcohol and Ether is an important topic included in the JEE syllabus for organic chemistry. Questions based on their reactions and methods of preparation are asked almost every year in JEE.
Frequently Asked Questions (FAQs)
Test to distinguish between alcohol and phenols
1. Litmus test: Phenols turn blue litmus red and alcohols have no effect.
2. Ferric chloride test: Phenols give blue, green, and violet colour while alcohol gives no change in colour.
3. Bromine water test: Phenols gives white precipitates while alcohol gives no precipitates.
A hydroxyl group is attached to a carbon atom of alcohols. —OH is attached to a benzene ring in phenols. (This class's "parent" molecule ule is also called phenol: PhOH or C6H5OH.) The ether functional group is formed when two carbon groups are connected to oxygen by single bonds.
Ortho-nitrophenol has a lower boiling point due to the formation of intramolecular H-bonding whereas p-nitrophenol forms intermolecular H-bonding.
Alcohols usually feature the hydroxyl group attached to aliphatic hydrocarbons. Phenols usually contain aromatic hydrocarbons. In comparison to phenol, alcohols are known to be less acidic. Phenols are relatively more acidic in nature and should, therefore, be diluted before usage.
The basic components used to make detergents, antiseptics, and perfumes are alcohols, phenols, and ethers, respectively.

