- The chemical formula of ethanoic acid is $\mathrm{CH}_3 \mathrm{COOH}$.
- It is a common household substance.
- A dilute aqueous solution of ethanoic acid (about 5–8%) is known as vinegar.
- Vinegar is widely used in cooking and food preservation.
- In laboratories, pure ethanoic acid is commonly used as a chemical reagent.
- During winter, ethanoic acid can freeze and form ice-like crystals.
- Due to its glacier-like appearance in solid form, it is called glacial acetic acid.
Ethanoic Acid - Overview, Structure, Properties & Uses, Preparation, FAQs
What gives vinegar its sharp smell and sour taste? Why does ethanoic acid play such an important role in our kitchens, laboratories, and industries? You will find these answers by reading this article on ethanoic acid. Ethanoic acid, commonly known as acetic acid, is an important organic compound widely found in everyday life and in chemical industries. It is best known as the main component of vinegar, which gives it a characteristic sour taste and pungent smell.
This Story also Contains
- Formula of Ethanoic Acid
- Structure of Ethanoic Acid:
- Properties of Ethanoic Acid
- Methods of Preparation of Ethanoic Acid
- Reactions of Ethanoic Acid
- Uses of Ethanoic Acid
- Some Solved Examples
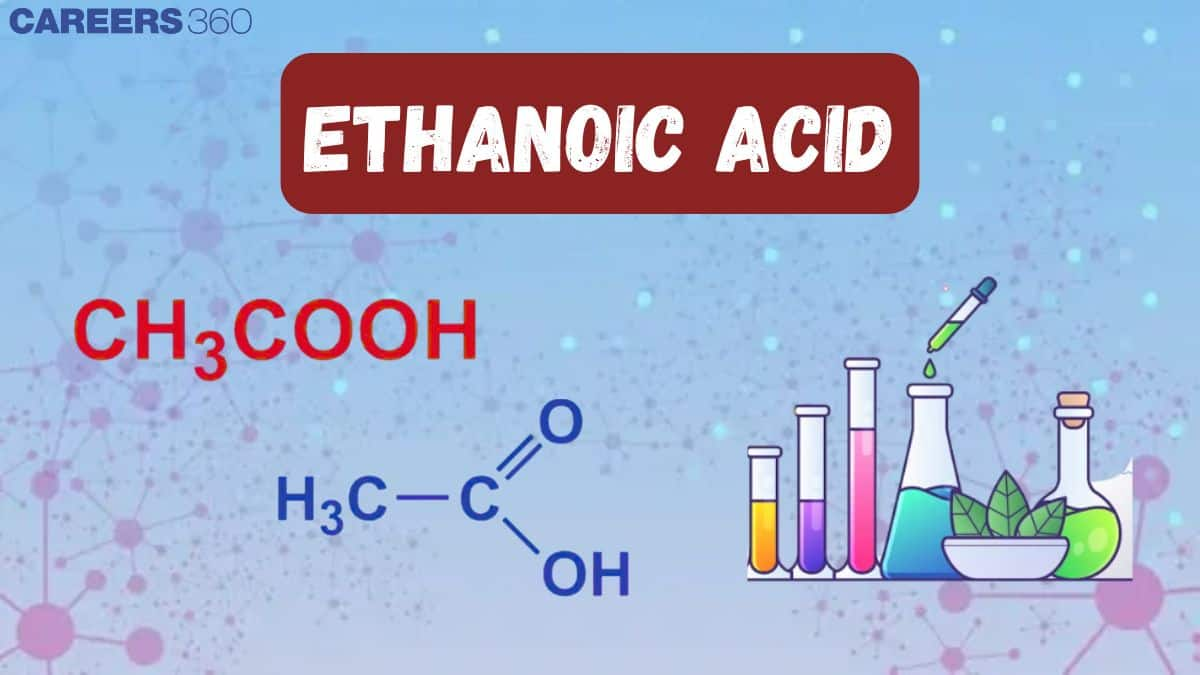
Formula of Ethanoic Acid
Ethanoic acid, commonly known as acetic acid, is a member of the carboxylic acid family. This family includes methanoic acid (a one-carbon carboxylic acid) and ethanoic acid, which contains two carbon atoms.
Structure of Ethanoic Acid:
- The structural formula of ethanoic acid is $\mathrm{CH}_3 \mathrm{COOH}$.
- Its molecular formula is $\mathrm{C}_2 \mathrm{H}_4 \mathrm{O}_2$.
- The molar mass of ethanoic acid is 60.05 g mol⁻¹.
- It consists of a methyl group $\left(-\mathrm{CH}_3\right)$ attached to a carboxylic acid group (–COOH).
- The carboxyl group contains a carbonyl group (C=O) and a hydroxyl group (–OH).
- The carbon atoms in the carboxyl group are $\mathrm{sp}^2$ hybridized.
- Ethanoic acid has a planar structure around the –COOH group.
- In liquid and vapour states, ethanoic acid molecules form dimers.
- Dimer formation occurs due to intermolecular hydrogen bonding between oxygen and hydrogen atoms.
Properties of Ethanoic Acid
Appearance: Ethanoic acid is a colourless, clear liquid with a sharp, pungent (vinegar-like) odour.
Melting Point: 16.6 °C; below this temperature, it solidifies, forming ice-like crystals (glacial acetic acid).
Boiling Point: 118.1 °C at atmospheric pressure.
Density: $1.05 \mathrm{~g} \mathrm{~cm}^{-3}$ at 25 °C.
Flash Point: Approximately 39 °C.
Solubility: Completely miscible with water in all proportions.
Solubility in Organic Solvents: Soluble in ethanol, ether, acetone, benzene, and glycerol.
Hydrogen Bonding: Forms intermolecular hydrogen bonds and exists as dimers in liquid and vapour states.
Vapour Pressure: About 15.7 mmHg at 25 °C.
pH: A 1.0 M solution of ethanoic acid has a pH of about 2.4.
pKa Value: 4.76 at 25 °C, indicating weak acidic nature.
Storage Conditions: Stable under normal laboratory conditions and stored in tightly closed containers.
|
Related Topics link, |
Methods of Preparation of Ethanoic Acid
1. Oxidation of Ethanol
Ethanoic acid is commonly prepared by the oxidation of ethanol using oxidising agents such as acidified potassium dichromate $\left(\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7\right)$ or potassium permanganate $\left(\mathrm{KMnO}_4\right)$.
$\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{OH}+[\mathrm{O}] \rightarrow \mathrm{CH}_3 \mathrm{COOH}+\mathrm{H}_2 \mathrm{O}$
- Ethanol is first oxidised to ethanal and then to ethanoic acid.
- This method is widely used in laboratories.
2. From Vinegar (Fermentation Method)
In nature and industry, ethanoic acid is produced by the fermentation of sugars.
$\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}+\mathrm{O}_2 \xrightarrow{\text { Acetobacter }} \mathrm{CH}_3 \mathrm{COOH}+\mathrm{H}_2 \mathrm{O}$
- Ethanol is oxidised by Acetobacter bacteria in the presence of air.
- This process produces vinegar, which contains $5-8 \%$ ethanoic acid.
3. From Methyl Cyanide
Ethanoic acid can be prepared by the hydrolysis of methyl cyanide (acetonitrile).
$\mathrm{CH}_3 \mathrm{CN}+2 \mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{CH}_3 \mathrm{COOH}+\mathrm{NH}_3$
Related Topics Link
Reactions of Ethanoic Acid
Esterification Reaction
Carboxylic acids react with alcohols in the presence of a catalyst (usually concentrated sulphuric acid) to form esters. This reaction is known as esterification.
Example:
When ethanoic acid reacts with ethanol, it forms ethyl ethanoate, an ester with a pleasant fruity smell.
$\mathrm{CH}_3 \mathrm{COOH}+\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{OH} \rightarrow \mathrm{CH}_3 \mathrm{COOCH}_2 \mathrm{CH}_3+\mathrm{H}_2 \mathrm{O}$
Saponification Reaction
Esters react with strong bases (alkalis) such as sodium hydroxide to form soap and alcohol. This reaction is called saponification and is commonly carried out using higher molecular mass esters (fats and oils).
$\mathrm{RCOOR}^{\prime}+\mathrm{NaOH} \rightarrow \mathrm{RCOONa}+\mathrm{R}^{\prime} \mathrm{OH}$
- Ester + Sodium hydroxide → Soap (sodium salt of fatty acid) + Alcohol
- This reaction forms the basis of soap manufacturing.
Reaction with Bases (Neutralisation)
Ethanoic acid, being a weak acid, reacts with bases to form a salt and water.
$\mathrm{CH}_3 \mathrm{COOH}+\mathrm{NaOH} \rightarrow \mathrm{CH}_3 \mathrm{COONa}+\mathrm{H}_2 \mathrm{O}$
- Ethanoic acid + Sodium hydroxide → Sodium ethanoate + Water
- Sodium ethanoate is used in the textile industry, food preservation, flavouring, and as a buffer solution along with ethanoic acid.
Formation of Carboxylic Acid Derivatives
Ethanoic acid can form various carboxylic acid derivatives by reacting with different reagents. These derivatives include:
- Acid chlorides
- Acid anhydrides
- Esters
- Amides
These compounds form an important class of organic chemicals and are widely used in industrial and laboratory processes.
Uses of Ethanoic Acid
- It is used in the production of synthetic polymers and vinegar.
- Used as vinegar (5–8% aqueous solution) for food preservation, pickling, and flavouring.
- Acts as a preservative and acidity regulator
- Ethanoic acid is used as a lysing agent for blood cells in laboratory and medical applications.
- It is used to inhibit the growth of fungi and acts as an antifungal substance.
Also read -
Some Solved Examples
Question 1: Ethanoic acid exists as dimers in liquid and vapour phases mainly due to:
A. Covalent bonding
B. Van der Waals forces
C. Intramolecular hydrogen bonding
D. Intermolecular hydrogen bonding
Solution:
Two ethanoic acid molecules form dimers via intermolecular hydrogen bonding between the –OH hydrogen and the carbonyl oxygen.
Hence, the correct answer is option (D)
Question 2: The hybridisation of the carbon atoms present in the -COOH group of ethanoic acid is:
A. $s p^3$
B. $\mathrm{sp}^2$
C. $s p$
D. One $s p^2$ and one $s p^3$
Solution:
Both carbon atoms in the carboxyl group are sp² hybridised.
Hence, the correct answer is option (B)
Question 3: Which of the following statements about ethanoic acid is incorrect?
A. It has a melting point of about 16.6°C
B. It is miscible with water in all proportions
C. It has a fruity smell in pure form
D. It is called glacial acetic acid in the solid state
Solution :
Fruity smell is a property of esters, not ethanoic acid.
Hence, the correct answer is option (C)
Question 4: Ethanoic acid reacts with sodium carbonate to form:
A. Sodium ethanoate, hydrogen, water
B. Sodium ethanoate, CO₂, water
C. Sodium acetate, oxygen, water
D. Sodium acetate only
Solution:
$2 \mathrm{CH}_3 \mathrm{COOH}+\mathrm{Na}_2 \mathrm{CO}_3 \rightarrow 2 \mathrm{CH}_3 \mathrm{COONa}+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}$
Hence, the correct answer is option (B)
Practice more questions with the link given below:
Frequently Asked Questions (FAQs)
As part of printing processes, acetates like metal and vinyl are used; cellulose acetate, used in textiles and photographic films, is made of acetate; and volatile organic esters are made from acetate.
Salad dressings are not the only application for acetic acid. Various foods are preserved with this ingredient as natural preservatives and antibacterial, such as vegetable pickles and edible films.
Acetic acid, also known as ethanoic acid, is simply ethanoic acid diluted in water. The majority of ethanoic acid is now synthesized and used in the production of plastics and other fundamental materials.
Weak acids, such as ethanoic acid (acetic acid), are those in which their hydrogen has not been completely released into water. This results in a partial dissociation and equilibrium with its conjugate base.
Acetic acid and ethanol produce a sweet aroma with their ester. Esters interact modestly with each other. A smaller intermolecular force of attraction makes ester compounds volatile. - Esters do not have hydrogen bonds.

